Photonics Products: Raman Spectrometers: Emphasis on biosciences
In Raman spectroscopy, laser light is inelastically scattered from the material under test. The resulting small change in wavelength of the scattered light, to a longer wavelength (Stokes shift) or shorter wavelength (anti-Stokes shift), is caused by a light-induced change in a vibrational or rotational state of the molecule from which the light is Raman-scattered.
Because of the unique spectral signatures of a vast number of organic molecules, Raman spectroscopy is widely used in medicine, the biosciences, and the food and drug industries to identify and characterize organic substances. Raman spectrometers can be readily integrated with microscopes to provide high-resolution data, even for in vivo science and medicine.
A wide variety of Raman spectroscopy products for bioscience and medicine is available on the market. While this article cannot include more than a small portion of the available products, a representative sampling of what is available is presented here.
Dispersive 1064 Raman spectroscopy for biosciences
“Raman proves to be by far the most powerful analytical technique for many applications, with high specificity, high accuracy, and ideally nondestructive, direct measurement,” says Lynn Chandler, spectroscopy product manager at BaySpec (San Jose, CA). “Owing to technological improvements spurred on by the telecommunications boom of the last decade, Raman spectroscopy has become much more accessible to users in all fields. The combination of improved technology and the technique’s molecular sensitivity have led to a surge in Raman usage in a myriad of application areas, especially in biosciences. In all these Raman applications, however, there remains a struggle to extract useful Raman spectra from complex biometrics often interfered with fluorescent and luminescent background.”
Fluorescence is much more likely and intense at short wavelengths where energies are more apt to cause an electronic transition, Chandler explains. So, despite the lower Raman scattering cross-section (inversely proportional to the fourth power of the wavelength), most users with potentially fluorescent samples long ago switched to near-infrared (near-IR) wavelengths such as 785 or 830 nm.
“These laser wavelengths push the detected signal to the spectral edge of silicon used in conventional multichannel CCD detectors, which are largely transparent above about 1050 nm,” Chandler says. “Yet even at these near-IR wavelengths, many substances still fluoresce, sometimes prohibiting Raman spectral acquisition. For those users who require longer wavelengths such as 1064 nm, the only available option has been Fourier-transform (FT) Raman, which is typically noisier and slower than dispersive Raman systems.”
Chandler says that the dispersive 1064 nm Raman spectrometer family of instruments developed initially by BaySpec (see Fig. 1) are handheld devices that combine the speed, sensitivity, and rugged design of traditional dispersive Raman instruments with fluorescence avoidance. Examples include handheld 1064 Raman for direct sampling (released in 2010 by BaySpec, the handheld Raman product line was sold and transferred to Rigaku Raman Technology in 2011); a fiber-optic probe for sampling (released in 2012 by BaySpec, the Agility Raman product line was sold and transferred to Anton Paar in 2016); and a 1064 confocal Raman microscope (released in 2012 by BaySpec, the Nomadic Raman line was sold and transferred to Anton Paar in 2016).
Conventional FT-Raman with excitation at 1064 nm is not compatible with confocal optics, and FT-Raman microscope systems have limited spatial resolution of around 50 µm. In contrast, Chandler says, the dispersive 1064 nm Nomadic Raman system permits diffraction-limited optical performance, allowing confocal microscopic Raman at 1064 nm to achieve diffraction-limited spatial resolution of around 1 µm. Such a confocal system will find wide applications in many areas, especially when microanalysis is required for biological sample analysis, Chandler says.
In one example of its use, a 1064 nm confocal Raman microscope was used to sample highly fluorescent amber, overcoming the challenge of high fluorescence and meeting the requirement for a 2 µm spot size.1 “Only 1064 dispersive Raman is capable of this,” Chandler says, who notes that this technique will provide a viable way for the analysis of complex biological samples noninvasively with selectivity and sensitivity, allowing deep penetration within the tissue for fluorescence avoidance.
The importance of aberration-corrected optics
“Raman spectroscopy in biological tissues often uses excitation lasers at high wavelengths and detection in the near-IR (due to autofluorescence),” notes Sebastian Remi, applications scientist at Princeton Instruments (Acton, MA). “It is explored/used for various applications in biosensing2, 5 and clinical diagnostics (cancer detection).3, 4 The technical challenge in these applications is often to reduce autofluorescence (by detecting in the near-IR range), and optimize system efficiency and throughput (high-throughput optics, sensitive detectors in near-IR) to increase signal-to-noise (SNR) and enable fast observation times for in vivo and clinical measurements to aid patient comfort. Applications often involve fiber-optic probes that are optimized for the specific application and allow for in vivo observations in patients.”
Princeton Instruments’ LS-785 system (see Fig. 2), together with its BLAZE or PIXIS cameras, has specs to optimize this process, Remi says. It has a high input aperture that matches the aperture of fiber-optic probes so that there is no light loss. The system uses lens optics with near-IR-optimized antireflection coatings and gold-coated gratings to optimize throughput. Back-illuminated deep-depleted cameras have high efficiency in the near-IR. PIXIS and BLAZE cameras have high cooling to minimize the dark noise for Raman applications for highest SNR, according to Remi. One sensor option on the BLAZE camera uses a super-deep-depleted high-resolution sensor that at least triples efficiency in the near-IR.As described by Remi, a compact spectroscopy system made by Princeton Instruments called FERGIE has aberration-corrected optics and a deep-cooled high-efficiency CCD detector. The technical design is optimized for ease of use, so researchers with nonspectroscopy backgrounds can perform complex Raman measurements quickly. FERGIE cubes are self-aligning optical elements that can be connected to FERGIE systems to configure it for different spectroscopic tasks.
Another system, the IsoPlane with PIXIS/BLAZE cameras, has optical designs that eliminate/minimize most optical system aberrations, increasing resolution, SNR, and system throughput, Remi says. Distortion-free optical imaging is also required for multispectral and hyperspectral Raman applications such as microspectroscopy or imaging of fiber bundles.
In one example of medical use of the LS-785 spectrometer, researchers from Vanderbilt University (Nashville, TN) evaluated a process to use Raman spectroscopy for fast assessment of breast-cancer surgery.3 The ability to collect signal from a fiber bundle with a large number of fibers, high imaging power, and high efficiency allowed the researchers to collect Raman “spectra with the same SNR […] 4X faster” than in previous experiments.
Bacteria detection
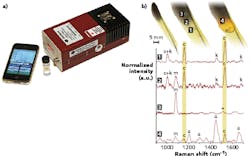
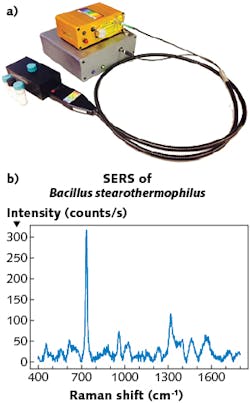
“The development of field-deployable, rapid, and affordable sensors for different types of bacteria detection would help across many sectors of bioscience,” says Jason Pierce, director of business development, instrument sales, and support at StellarNet (Tampa, FL). “For example, StellarNet portable systems have been used in conjunction with different surface-enhanced Raman spectroscopy (SERS) technologies to detect both bacillus stearothermophilus and Escherichia coli (see Fig. 3). This technology can be deployed to many food-distribution centers to identify and reduce contamination.”StellarNet produces a variety of high-performance modular Raman spectrometer systems, Pierce notes. The company offers “just about every Raman wavelength from 405, 532, 633, 785, 830, and 1064 nm as standard options,” he says. The combination of small size and high performance allows for a research-grade system to be easily transported from clinic to laboratory, and the systems can easily couple to a microscope or used with SERS techniques to amplify signal.
The StellarNet Raman-HR-TEC spectrometer has a variety of cooling options to boost performance. For example, a one- or two-stage thermoelectric cooler (TEC) for the detector can be chosen, which reduces noise 60–90% at long exposure times. The spectrometer has exposure time capabilities of up to 8 minutes—important for many biological applications where low-power lasers are required so as to not damage samples, Pierce says.
Raman assists surgeons in real time
Martin Isabelle, application scientist at Renishaw (Wotton-under-Edge, England), says applications of the company’s inVia Raman microscope systems across the globe have been quite diverse: examples include imaging intracellular processes to tissue diagnostics with the ability to discriminate between cancerous and normal tissue, and analyzing the hydathode pits in alpine plant leaves to understand leaf development in plant biology.
“Our Raman microscopes are available in both upright and inverted configuration, and can be connected to complementary techniques such as conventional fluorescence or laser scanning confocal microscopy,” Isabelle says. “These systems have a range of laser wavelengths and technologies including focus tracking and ‘Streamline,’ which collects high-quality data using a laser line rather than a spot to avoid photothermal damage to delicate biological samples and provide faster acquisition of spectra.”
Renishaw has also recently introduced a benchtop Raman imaging instrument specifically for biological analysis, Isabelle notes. The system uses a 785 nm wavelength laser and is designed specifically for scientists who are translating their research into clinical applications. With an emphasis on tissue and biopsy samples, the system is designed to be easy to use and suitable for use within a clinical setting.
The company’s RA814 instrument (see Fig. 4) is a compact Raman imaging system with both low (macro) and high-magnification microscopic imaging capability, which enables users to view the context of a biopsy or tissue slide using the macro view, and select regions for microscopic and Raman analysis with the inbuilt 785 nm laser. The software includes data analysis to enable generation of pathology predictive models of disease. Slated to be launched soon commercially, the RA814 is a variant of Renishaw’s RA802 pharmaceutical instrument already on the market.“The system has inbuilt reference calibration and has been developed to offer a level of repeatability between instruments to allow these predictive models to be transferred from one instrument to the next,” Isabelle says. “The validation of this was part of a UK-funded project that involved three separate hospital and university-based institutions investigating the ability of the instrument to diagnose gastrointestinal (GI)-tract cancers and transfer the model between operators, instruments and locations. To our knowledge, this is the first project of this kind which is critical to the adoption of the technique to a robust clinical solution.”7, 8
In an example, an RA814 compact Raman imaging system is used in neuro-oncology, with systems installed at Oxford (England) Radcliffe hospital as well as at the Neuro-center of Humanitas Research Hospital (Milan, Italy). The systems are situated next to the day surgical theaters to allow research staff to collect brain biopsy tissue during surgical operations and analyze the tissue in real time, providing intraoperative diagnostic evaluation of the tissue pathology (cancer or noncancerous) to support the optimal removal of brain cancer tissue. In addition, the system supports the pathology department, providing postoperative assessment of tissue biopsies.
Systems and substrates for SERS
Ocean Optics (Dunedin, FL) produces a range of modular Raman spectroscopy products that can be combined in multiple configurations to create a complete Raman system. These are used for measurements across a range of application areas, including biomedical, life science, materials characterization, pharmaceutical, forensics, security, safety, and process control. Raman’s extreme specificity and relative ease of use make it ideal for identifying unknown samples or materials, driving its popularity for a range of application areas and markets, according to two of the company’s scientists, Anne-Marie Dowgiallo, sensor R&D scientist, and Yvette Mattley, senior applications scientist.
Any of the company’s Raman spectrometers can be used for biomedical applications, including the QE Pro-Raman (near-IR version also available, called the NIRQuest) and bundled versions available for 532, 638, 785, and 1064 nm excitation, called the Maya2000 Pro (near-IR version also available).
The QE Pro high-sensitivity spectrometer (see Fig. 5), which is designed for low-light-level measurements such as Raman and fluorescence, has a sensitive, high-quantum-efficiency, back-thinned detector coupled with a thermoelectric cooler to enable long measurement times without thermal noise overwhelming the low Raman signals, according to Ocean Optics. The detector can be cooled to 40°C below ambient temperature, resulting in a stable baseline and low noise while acquiring data for integration times up to 60 minutes. The QE Pro-Raman is preconfigured for measurements using 785 nm Raman excitation, but can be configured to cover any excitation wavelength across the UV-VIS-NIR range.Biological samples present challenges for Raman measurements, including a high-fluorescence-background component, the fact that Raman is limited primarily to the surface of the optically dense biological samples like tissue with a shallow sampling depth and small sample area, and, for clinical measurements, long measurement times with powerful lasers, making it less than ideal for biological measurements of tissue or other optically dense samples.
As described by the two Ocean Optics scientists, to take advantage of the powerful specificity of Raman measurements, many novel techniques and custom measurement configurations have been developed to overcome the limitations of Raman spectroscopy of biological samples. Sophisticated background correction and multivariate analysis are also used to extract meaningful data from complicated Raman spectra of complex biological samples like tissue. For Raman signal enhancement, Ocean Optics provide a line of SERS substrates that are based on gold or silver nanoparticles.
Examples of the use of SERS include the detection of proteins, DNA, RNA, pathogens, and even live cells. Detection techniques typically fall into two categories: one involves the biomolecule adsorbing to the surface of a nanoparticle or nanostructured metallic film, and the other involves using a Raman dye or label to indirectly detect the biomolecule. Two examples are given (in both examples, the instrumentation is from Ocean Optics, although the SERS substrates aren’t).
First, an exogenous cancer biomarker, acetyl amantadine (AcAm), was detected in urine solutions using SERS.9 The Raman experiments were performed with a 785 nm fiber-coupled laser diode (23 mW), a QE 65 Pro portable spectrometer, and a fiber-optic probe responsible for excitation, collection, and filtering. The authors favored this low-cost setup that can be used for the longer integration times typically required for Raman spectroscopy. This experimental setup coupled with commercial SERS substrates is promising for clinical adoption for early cancer detection as a low-cost and rapid tool.
Second, SERS was used to enable protein detection and recognition for applications targeting protein-protein interactions for the field of proteomics.10 The SERS substrate was coated to specifically bind bovine serum albumin (BSA), a common protein studied for biological systems. SERS experiments were performed with the QE Pro configured for 785 nm laser excitation at a power of 12 mW, a grating of 1200 lines/mm, and an input slit of 50 µm. The spectra were collected in the range between 600 and 1800 cm-1 with 10 s integration time. The Raman system was coupled with an Olympus BX51 microscope with a 50X (NA = 0.75) objective. This demonstrates how Ocean Optics hardware can be integrated into a microscope configuration for biomedical applications.
REFERENCES
1. D. B. Thomas et al., Sci. Rep., 4, 5226 (2014).
2. A. Moody et al., Anal. Chem., 89, 11, 5688 (2017); http://dx.doi.org/10.1021/acs.analchem.7b00985.
3. G. Thomas et al., Sci. Rep., 7, 13548 (2017); http://dx.doi.org/10.1038/s41598-017-13237-y.
4. K. Lin et al., Theranostics, 7, 3517 (2017).
5. C. Hanson et al., Sensors, 17, 327 (2017).
6. C. E. Johnson et al., J. Forensic Sci., 63, 1, 66–79 (Jan. 2018).
7. K. Lau et al., “The road map towards providing a robust Raman spectroscopy-based cancer diagnostic platform and integration into clinic,” Proc. SPIE, 9704, 97040B (2016); doi:10.1117/12.2230007.
8. M. Isabelle et al., Faraday Discuss., 187 (2016); doi:10.1039/c5fd00183h.
9. G. Cao et al., Analyst, 139, 5375–5378 (2014).
10. M. Rippa et al., Opt. Data Process. Storage, 3, 54–60 (2017).
For More Information
Companies mentioned in this article include:
BaySpec
San Jose, CA
www.bayspec.com
Ocean Optics
Dunedin, FL
www.oceanoptics.com
Renishaw
Wotton-under-Edge, England
www.renishaw.com
Princeton Instruments
Acton, MA
www.princetoninstruments.com
StellarNet
Tampa, FL
www.stellarnet.us
DISCLAIMER: While we try to include information from the broadest possible number of companies that manufacture the products featured in our Photonics Products series, because of limited word count as well as deadlines that cannot always be met by requested contributors, we cannot possibly include all companies and regret if your company is not included in our series.
About the Author
John Wallace
Senior Technical Editor (1998-2022)
John Wallace was with Laser Focus World for nearly 25 years, retiring in late June 2022. He obtained a bachelor's degree in mechanical engineering and physics at Rutgers University and a master's in optical engineering at the University of Rochester. Before becoming an editor, John worked as an engineer at RCA, Exxon, Eastman Kodak, and GCA Corporation.