VIBRATIONAL PHOTOACOUSTIC MICROSCOPY: Converting molecular vibration to mechanical for deep-tissue imaging
Jie Hui and Ji-Xin Cheng
Enabling the detection of specific molecules within cells and tissues, molecular vibration is the basis of numerous microscopy approaches. Among these are Raman scattering and infrared absorption vibrational microscopy, which have been widely used for chemical imaging in biomedicine. Similarly, such nonlinear vibrational methods as coherent anti-Stokes Raman scattering (CARS) and stimulated Raman scattering (SRS) microscopy have enabled new discoveries in biomedical imaging, thanks to high sensitivity and 3D spatial resolution. But because all these approaches are limited to an imaging depth of a few hundred micrometers in biological tissue, their potential in clinical settings is restricted.
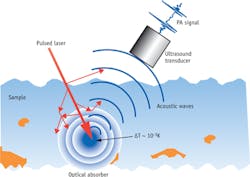
A deep-tissue imaging approach able to maintain high chemical selectivity and spatial resolution would certainly satisfy diagnostic needs in biomedicine. A promising solution is a photoacoustic (PA) imaging platform combining light illumination with acoustic detection based on the PA effect discovered by Alexander Graham Bell as early in 1880.1 The PA signal can be generated by either microwave or laser pulses. In PA imaging, a pulsed laser beam illuminates a specimen, which contains optical absorbers with significant absorption at the laser wavelength. After much scattering in tissue, the diffused photons are delivered to and absorbed by the target molecules. Part of the energy is converted to heat in the region of the absorbers, which results in a local temperature rise on the order of millikelvins and the subsequent generation of acoustic waves. The acoustic waves then can be recorded by an ultrasound transducer in real time. The time-of-flight of the PA signal contains information about the location of the absorbers, so the distribution of the absorber can be reconstructed from the measured signal (see Fig. 1).
Photoacoustic advantages
The PA imaging technique offers several unique advantages over its pure optical counterparts. First, it can achieve higher resolution in deep tissue based on the detection of acoustic waves, as acoustic scattering in tissue is one thousand-fold weaker than the optical scattering. Second, because the PA signal originates from absorption of photons, it eliminates the background caused by scattering and offers a molecular contrast based on optical absorption. Third, the generation of PA signal does not depend solely on ballistic photons; diffused photons equally contribute to the signal, which leads to a centimeter-scale imaging depth in a tomography configuration.
Taking advantage of these advantages, researchers have developed various PA imaging platforms, including photoacoustic microscopy (PAM), photoacoustic tomography (PAT), and intravascular photoacoustic imaging (IVPA). Besides PA imaging of hemoglobin, exogenous PA contrast agents such as dyes and nanoparticles have been intensively reported over the years. Wang and Hu described the various approaches in a comprehensive review of PA imaging.2
New contrast mechanism
In 2007, our laboratory at Purdue University published a theoretical study to explain our unexpected observation of Raman-resonance induced photodamage in CARS imaging.3 We found that with tight focusing, about 0.01 percent of the excitation energy is transferred to the specimen via SRS processes that occur simultaneously with CARS. Because molecular absorption could produce an ultrasound signal for deep-tissue imaging, as shown in photoacoustic detection of hemoglobin by others, we began to explore whether we could "hear" the SRS with a transducer. We spent two years, but no ultrasound signal was detected under the tissue photodamage threshold.
Although frustrated, we decided to try once more by using aqueous carbon disulfide (CS2), a molecule known to produce a strong signal of Raman-induced Kerr effect. We scanned the optical parametric oscillator (OPO) idler wavelength to search for the Raman-stimulated PA signal and found a strong ultrasound spike at 850 nm for the OPO and 1064 nm for the YAG. Surprisingly, the wavenumber difference of the two excitation beams did not match with any Raman band of CS2. Moreover, the signal remained when blocking the YAG laser. We immediately realized that we were observing a completely new process that is likely based on overtone excitation of CS2 vibration. This finding led to our development of vibrational photoacoustic (VPA) microscopy.4 Using the second overtone of the C-H bond, we demonstrated 3D VPA imaging of lipid-rich atherosclerotic plaques by excitation from the artery lumen.
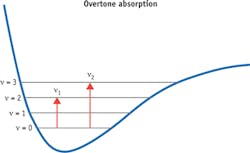
According to anharmonicity theory, the transition frequency for an overtone band has the following relation with the fundamental frequency,
v= v0n - Χv0(n + n2)
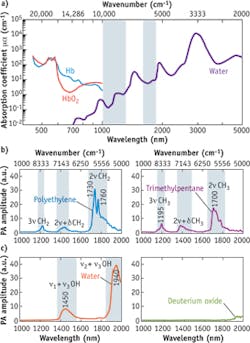
Electronic absorption of hemoglobin is dominant in an optical window ranging from the visible to the NIR region between 400 nm and 1.0 μm (see Fig. 4a). Hemoglobin absorption overwhelms the third- and higher-order C-H overtone transitions located in the same region. For wavelengths longer than 1.0 μm, significant water absorption due to overtone transitions of O-H bonds reduces penetration of the photons in the specimens. Nevertheless, two optical windows exist in the water vibrational absorption spectrum, 1000−1300 nm and 1600−1850 nm (highlighted in light blue). Importantly, the second and first overtone transitions of C-H bonds are located in the same windows. These spectral features produce two optical windows for C-H bond-selective imaging.
P. Wang et al. performed a detailed analysis of VPA spectra of C-H, O-H, and O-D bonds.5 They demonstrated that, as shown in Fig. 4b, "polyethylene provides the absorption profile of the methylene group (CH2), for which the first overtone (2ν CH2) shows two primary peaks at around 1730 nm (5800 cm-1) and 1760 nm (5680 cm-1). The stronger peak at 1730 nm is generally thought to be a combination band of asymmetric and symmetric stretching. The 1760 nm peak is assigned to the first overtone of stretching. The second combination band of CH2, located between 1350 and 1500 nm, is attributed to the combination of harmonic stretching and a non-stretching mode, such as bending, twisting, and rocking (2ν + δ). The second overtone of CH2 stretching is peaked at around 1210 nm. It was shown that the PA amplitude at 1730 nm is around 6.3 times that at 1210 nm. The spectrum of trimethylpentane is mainly contributed to by the absorption profile of methyl groups (CH3). The primary peak at around 1700 nm (5880 cm-1) is assigned to the first overtone of CH3 stretching."
The researchers further report, "It is clear that the CH2 and CH3 groups have distinguishable profiles at the first overtone region. The second combination band of CH3 starts from 1350 to 1500 nm, with the main peak at around 1380 nm, which is generally thought to be 2ν + δ. The second overtone of CH3 stretching shows the primary peak at around 1195 nm. Fig. 4c shows the VPA spectra of H20 and D20 liquid in the 1.0−2.0 μm window. The v2 + v3 combinational band and the v1 + v3 combinational band appear at 1940 and 1450 nm, respectively. Here, v1, v2, and v3 denote the symmetric, bending, and asymmetric vibrations of the water molecule, respectively. Due to the heavier mass of deuterium, the prominent overtone and combinational bands of D20 are located at longer wavelengths. Thus, D20 can be used as an acoustic coupling agent during vibration-based PA imaging."
A couple of applications can demonstrate the technique's advantages for biomedicine.
Mapping lipid-laden plaques
Lipid deposition is a major hallmark of atherosclerosis, and one that determines lesion progression and plaque vulnerability to rupture. Accurate characterization of the lipid content in an arterial wall would enable a phenomenal improvement for vascular intervention in diagnosis and treatment of atherosclerosis.
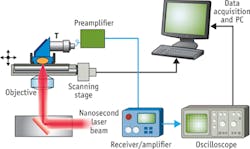
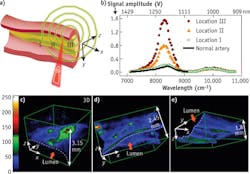
H.W. Wang et al. performed 3D label-free VPA imaging of lipid-rich atherosclerotic plaques of carotid arteries in the optical window of 1000 nm to 1300 nm.4 The arteries were harvested from Ossabaw pigs having metabolic syndrome and profound atherosclerosis. The imaging configuration is shown in Fig. 5a with optical excitation from the lumen side of the artery. VPA spectroscopy at different sites of atherosclerotic arterial walls has demonstrated clearly the capability of imaging different levels of lipid deposition inside arterial walls (see Fig. 5b). Locations I-III correspond to a thickened intima, an intermediate plaque without a necrotic core or fibrotic lesion, and a relatively advanced lesion with the formation of a lipid core, respectively. According to the VPA spectra of the lipid depositions, we used excitation laser light at 1195 nm for 3D VPA imaging of atherosclerotic lipid deposition for optimal vibrational contrast. Our images elucidate the spectra of the different milieus of lipid accumulation in the arterial wall, such as a confluent lipid core in an atherosclerotic artery, a scattered lipid deposition in an arterial wall, and the formation of mild fatty streaks in early atheroma (see Fig. 5c-e). A strong VPA signal from lipids located at 1.5 mm can be detected below the lumen.Beyond lab-based microscopy studies of lipid-laden plaques inside an atherosclerotic artery, VPA imaging can be applied clinically for diagnosis of cardiovascular disease (CVD). CVD, the number-one killer in the United States, results mainly from vulnerable plaques. Current imaging tools such as x-ray angiography, magnetic resonance imaging (MRI), and intravascular ultrasound (IVUS) lack chemical contrast, while optical coherence tomography (OCT) is limited by its imaging depth. Thanks to its chemical selectivity and millimeter-deep resolvability, catheter-based intravascular VPA (IVPA) can characterize atherosclerotic plaques in a clinical setting to meet this critical need for prognosis, diagnosis, and treatment assessment. Based on the overtone absorption of NIR light, it will be a game-changer for diagnosis of CVD in the near future.
Assessing white matter loss, regeneration
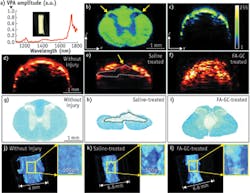
VPA imaging is also a valuable label-free imaging tool for assessing the progression of white matter pathology after spinal cord injury (SCI) in a rat model as well as the treatment effects of neuroprotective therapeutics.
As reported by W. Wu et al., VPA spectroscopy was performed to determine its ability to characterize the histology of a normal rat spinal cord and to search the optimal wavelength for VPA imaging of the white matter as shown in Fig. 6a.6CH2-rich white matter has two peaks located at 1730 nm for the first overtone and 1200 nm for the second overtone of CH2 vibration with the peak amplitude at 1730 nm, 5.8 times higher than that at 1200 nm (which is consistent with previous findings). The researchers performed C-scan VPA imaging of cross-sectional slices of a rat spinal cord to check the contrast difference between white matter and grey matter under 1730 nm excitation, which is likely caused by higher CH2 bond density in white matter (see Fig. 6b). They performed B-scan VPA imaging of fixed intact spinal cord under 1730 nm excitation, as shown in Fig. 6c, with 200 μm penetration depth for white matter and 1.3 mm at the dorsolateral surface of the cord above the dorsal horn. The higher scattering coefficient of white matter (296 cm-1 at 1064 nm) over gray matter (57 cm-1 at 1064 nm) explains the penetration difference. These results have suggested that the high VPA contrast and high scattering coefficient represent a unique feature of white matter in VPA images, allowing us to examine white matter morphology and changes in the normal and injured spinal cords.The researchers compared VPA images showing normal and injured spinal cords to determine whether VPA is a sensitive measure of pathological changes (see Figs. 6d and e). Further, they evaluated a neuroprotective treatment with ferulic acid conjugated glycol chitosan (FA-GC) to determine whether VPA imaging is sensitive for assessing the neuroprotective effect of a treatment agent (see Fig. 6f). In the B-scan VPA cross-section image of the injured spinal cord, the dorsal portion of the cord showed two times lower VPA signal compared to the normal spinal cord, indicating lower CH2 bond density.
Histology with Luxol fast blue staining (see Fig. 6g-i) highlighted an obvious tissue loss in the dorsal white matter at the injury site, which is consistent with the VPA image. The tissue loss may be attributed to myelin degeneration as a result of secondary injury. In the VPA image of FA-GC treated spinal cord (see Fig. 6f), there was no abrupt VPA signal loss on the dorsal surface of the spinal cord, indicating white matter recovery after the treatment. The 3D VPA image further confirmed our findings and provided us with additional information on the dimensions of the injury (see Figs. 6j and k) and FA-GC treatment-mediated recovery (see Fig. 6l).
Based on VPA imaging with 1730 nm excitation, where the first overtone vibration of CH2 bond is located, we can assess white matter loss after a contusive SCI in adult rats. Using the first overtone vibration of CH2 bond for contrast allowed label-free mapping of white matter in an intact spinal cord and observation of physiological changes before and after injury. Moreover, the recovery of the spinal cord from contusive injury with the treatment of a neuroprotective nanomedicine, FA-GC, was also observed.
Future promise
As a label-free imaging modality with chemical selectivity and millimeter-deep resolvability, vibrational photoacoustics provides a new avenue to map the chemical content of biological tissue. The broad applicability of this emerging technique will be demonstrated in the months and years to come. It will likely benefit research and shows great promise for clinical applications as well.
ACKNOWLEDGEMENTS
The authors cordially thank Dr. Lihong Wang and Dr. Michael Sturek for their collaborative effort in developing overtone vibrational transition as a new contrast mechanism for photoacoustic imaging.
REFERENCES
1. A. G. Bell, Am. J. Sci., 20, 118, 305−324 (1880).
2. L. V. Wang and S. Hu., Science, 335, 6075, 1458−1462 (2012).
3. H. Wang, Y. Fu, and J. X. Cheng, J. Opt. Soc. Am. B, 24, 3, 544−552 (2007).
4. H. W. Wang et al., Phys. Rev. Lett., 106, 23, 238106 (2011).
5. P. Wang, H. W. Wang, M. Sturek, and J. X. Cheng, J. Biophoton., 5, 1, 25−32 (2012).
6. W. Wu, P. Wang, J. X. Cheng, and X. M. Xu, J. Neurotrauma, doi:10.1089/neu.2014.3349 (2014).
JIE HUI, PhD candidate, is a teaching assistant in the Department of Physics, and JI-XIN CHENG, Ph.D., is a professor in the Weldon School of Biomedical Engineering and the Department of Chemistry, Purdue University (West Lafayette, IN; www.chem.purdue.edu/jcheng). Contact Dr. Cheng at [email protected].