Minimizing patient risk for light-based therapies
Light is increasingly becoming a promising tool in the fight against cancer. Although chemotherapy, surgery, and radiation therapy are widely known treatment methods, light-based treatments such as photodynamic therapy (PDT)1,2 and photoimmunotherapy (PIT)3 are growing in use.
PDT and PIT require homogeneous and efficient light-delivery systems, typically based on laser-light sources or LEDs, within a predefined power range. By taking the illumination source and attached light delivery system into account, a new sophisticated measurement approach is enabling more predictable and controlled (calibrated) light illumination, producing better treatment results in the clinical field.
Delivering high-quality light illumination
In many light-based therapy indications, light diffusers used at the distal end of a fiber-optic probe are important for homogenously spreading out the light beam so the tissue receives a predefined amount of light. For instance, when treating skin cancer with PDT, a front-emitting diffuser can apply light from outside the body. For tumors deeper inside the body, cylindrical diffusers ensure a homogeneous radiation pattern to activate the photosensitizer for PDT or PIT.
As light-based therapies evolve, there’s a growing need to deliver very accurate light intensities over a defined area—an important prerequisite for PDT and PIT applications. Advanced diffusers must transmit visible and near-infrared wavelengths—such as 980 nm or 1064 nm for laser interstitial thermal therapy (LITT)—with high temperature stability, homogeneity, and efficiency.
Creating a reliable light-delivery system requires extensive precision measurements of the light output characteristics. Although individual measurements can ensure a diffuser meets specifications, these measurements do not provide proof that the entire system—from the light source to the end-diffusing tip—performs optimally. This could result in a light power density too low to effectively activate the photosensitizer. Or it could be too high and “photobleach” the photosensitizer, which reduces the ability to kill cancer cells. In the worst case, it could thermally overheat human tissue.
Whole system measurements
Reliably managing the temperature of light illumination is crucial for minimizing patient risk during PDT and PIT. Activating the drug without scarring or scorching human tissue requires keeping the temperature below 42°C (almost 108°F). Given the nature of the glass material, diffusers, such as those developed by SCHOTT, absorb less light power and, therefore, do not heat up as much as other diffusers using other technologies.
It is also important to check for any thermal hot spots on the diffuser by using an infrared (IR) camera (see Fig. 1). These thermal images show temperatures routinely stay between 20°C and 35°C across the whole surface of the diffuser.
IR camera measurements are more reliable than other available methods. Contact measurement, for instance, does not capture temperature peaks and can misrepresent the actual surface temperature of a diffuser. Pyrometric approaches often integrate measurements from a large area, which can produce values lower than the actual temperature. These kinds of temperature measurement methods are well known for other state-of-the-art diffusers. However, they can be critical because they can misleadingly suggest lower temperatures than are actually in use. The potentially higher diffuser temperatures expose a high patient risk and might even lead to an unnecessary damage of the tissue.
Measuring optical performance
The relevant optical product characteristics of diffusers can each be determined in individual measurements and proven to be in accordance with the device maker’s spec requirements. However, the parameters are often measured and interpreted individually. Therefore, it is often not possible to predict the absolute outcome of the final product. This independent approach does not ensure the medical device system from the light source to the end-diffusing tip performs successfully or that the optimal level of power density will actually be applied in the treatment.
A holistic measurement system, developed by SCHOTT researchers, views all parameters in an aggregated way. The goal was to make it possible to calibrate the light power in the field with the application light sources to adjust the optimal optical power dose. This is done by measuring homogeneity and efficiency, and then mathematically combining the results, while also considering the calibration procedure of the application light source in the field.
Measuring homogeneity
Cylindrical diffusers used for PDT and PIT are manufactured from proprietary optical glass. Thanks to a customizable glass matrix that incorporates unique scattering elements, SCHOTT’s diffusers offer excellent homogeneous light radiation across the complete diffuser length, while avoiding peaks that could produce unwanted light emission hot spots. This enables surgeons to apply a virtually constant and highly reproducible light dose along the full length of the diffuser.
PDT and PIT applications require homogeneity within at least ±20%, while high performance devices typically show ±15% of the nominally intended illumination value. The homogeneity should ideally apply to the entire diffuser length with a deviation of max. 1 mm to 2 mm, depending on the total diffuser length. SCHOTT’s light output profile testing has shown high repeatability from batch to batch.
Evaluating images from a CCD camera is a reliable way to measure radiation homogeneity (see Fig. 2). This method offers valuable information on the tolerance range as well as the reproducibility among a batch of diffusers as a near-field measurement on the surface of the diffuser. It also allows for angle-dependent far-field measurements.Measuring efficiency
It is important for light diffusers to perform with the highest possible efficiency. For cylindrical diffusers, efficiency is high when emitting the light from the component’s circumference with as little light absorbed by the diffuser body as possible. Higher efficiencies produce less back reflection and less light energy absorbed. For PDT and PIT, a diffuser absorbing less light energy also stays cooler, which results in higher performance.
PDT and PIT applications usually require an efficiency of at least 70%, depending on the total diffuser length. Efficiency is measured by placing a diffuser within the center of a large integrating sphere (see Fig. 3).Putting it all together
Although efficiency measurements indicate how much light is irradiated to a specific area, they do not show how it is distributed across the whole object. Likewise, homogeneity of the light output is measured as a relational value, without the clear link to a specific scale. Only when efficiency and homogeneity are mathematically combined does it become possible to predict whether the light source will calibrate in the field with the correct output power or not.
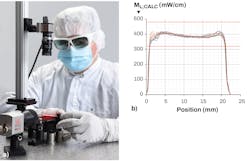
By combining diffuser homogeneity measurements with diffuser efficiency and pairing them with the calibration device of the light source in the field, it becomes possible to calculate the power radiated in relation to a surface area or body volume. This parameter, called radiant exitance or irradiation for superficial illumination, is usually measured in watts per square meter for frontal diffusers. For cylindrical diffusers, it is measured in milliwatts per centimeter of the diffuser length, which is called local radiant exitance or fluence rate. PDT and PIT applications typically require 150 mW/cm to 400 mW/cm, with an accepted deviation of ±20% (see Fig. 2b).
Process stability and system optimization
Most state-of-the-art diffusers available on the market are produced using scattering mechanisms such as surface roughness, concentration of scattering particles or the positioning of micro-scaled elements that are difficult to control and reproduce. In contrast, SCHOTT can design and manufacture the glass optical fibers used to make its diffusers in a way that every position on a fiber-based component will offer the same optical characteristics and performance. This includes consistency and reproducible optical performance across batches, avoiding axial or angular deviations. Thus, it might be possible in future to avoid—in next-level illumination systems—the current necessity of calibrating the light delivering diffusers in the treatment field with the light source.
REFERENCES
1. M. A. Biel, Adv. Exp. Med. Biol., 831, 119–136 (2015); doi:10.1007/978-3-319-09782-4_8.
2. F. J. Civantos et al., Adv Ther., 35, 3, 324–340 (Mar. 2018).
3. D. M. Cognetti et al., Head & Neck, 43, 12, 3875–3887 (Oct. 9, 2021).
About the Author
Haike Frank
Marketing Director, SCHOTT AG Lighting and Imaging
Haike Frank, Ph.D., is the Marketing Director at SCHOTT AG Lighting and Imaging (Mainz, Germany).
Hubertus Russert
Senior Development Engineer, SCHOTT AG Lighting and Imaging
Hubertus Russert is a senior development engineer at SCHOTT AG Lighting and Imaging (Mainz, Germany).
Bernd Schultheis
Manager of Application Engineering Medical, SCHOTT AG Lighting and Imaging
Bernd Schulthe is the Manager of Application Engineering Medical at SCHOTT AG Lighting and Imaging (Mainz, Germany).