Spectrometers: Excitation source parameters dictate Raman spectroscopy outcomes
In most spectroscopic techniques, the effects of the light source are easily decoupled from the measured spectroscopic data. For example, in traditional transmission, absorption, and reflection spectroscopy, the light source is typically a light bulb or glow bar powered by a constant current to minimize fluctuations in the optical output power.
Provided a minimum power fluctuation is maintained and the emission spectra are sufficiently broad to cover the area of interest, the absolute spectral irradiance is completely meaningless because once a reference spectra is taken (representing either 100% transmission or 100% reflection), it is used to self-normalize all of the subsequent data, producing a spectra devoid of any dependence on the illumination source.
Similarly, in fluorescence spectroscopy, the excitation source must simply contain a high-enough concentration of energy in the spectral region overlapping with the analyte's electronic absorption band. Depending on the analyte and system constraints, this can be done with anything from an unfiltered pulsed xenon flash lamp to a light-emitting diode (LED) or laser. As long as there is a stable and repeatable number of photons with the proper energy incident on the sample, the properties of the excitation source are completely decoupled from the resultant spectral data.
Raman spectroscopy, however, is unique among spectroscopic techniques for its extreme dependency on the properties of its excitation source. To understand the intricacies of this dependence, it is necessary to start with a fundamental physics review of the Raman effect.
The Raman effect
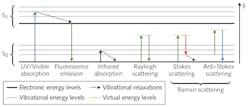
In simple terms, the Raman effect arises from phonon-photon interactions that can be represented in a Jablonski diagram similarly to fluorescence (see Fig. 1). The most notable difference between Raman and fluorescence is the fact that there is no required electronic transition, meaning the energy of the resultant photons is independent of the properties of the molecule. The photon's energy is instead dependent on the difference between the initial energy of the incident photon and the phonon energies of the vibrational modes of the molecule induced by the incident photon's electric field.
From this, we can see why the Raman effect is commonly defined as the inelastic scattering of light by molecules since the incident photon is never truly absorbed and, therefore, the resultant light is never truly emitted. Rather, the incident photon slams into the molecule, imparts or absorbs energy, and then scatters off at a different angle.
It is helpful to think of this process as the Compton effect on a molecular level.1 By illuminating the sample with a monochromatic (laser) source and collecting the scattered light and measuring the various shifts in wavelength, one can determine the different vibrational modes excited in the molecule and, therefore, the bond structure of the molecule itself.
The all-important laser
Even though the Raman effect was first presented by Sir C. V. Raman to the South Indian Science Association in March 1928, the technology of the time was not advanced enough for Raman spectroscopy to be a viable analytical technique.2 There were a few attempts to develop Raman instrumentation over the next 30 years, such as the Huet BII and the Cary 81, both of which used low-pressure mercury lamps as an excitation source. However, it wasn't until the invention of the helium neon (HeNe) laser in 1962 that Raman spectrometers first became truly viable.
The potential of the HeNe laser was almost immediately recognized and in 1964, PerkinElmer officially announced the world's first "laser Raman spectrometer" at an international spectroscopy symposium held at Ohio State University.3
Over the past 55 years, lasers have advanced considerably, but the spectral properties that made the HeNe the ideal source in 1962 are still relevant today—namely its spectral stability and narrow linewidth. Lasers are also desirable as an excitation source for Raman spectroscopy because of their directionality and focusability—critical attributes that require a closer look at the physics of the Raman effect to be understood.
Because a detailed quantum mechanics derivation of Raman scattering is beyond the scope of this article, we will skip to the end result:
P α σR Iο
Here, P is the power of the Raman scattered light, σR is the Raman scattering cross-section, and Io is the intensity of the light on the material.4 The ability to tightly focus the incident light on the sample is extremely important because of the dependency on the intensity—not just the power—of the excitation source. From a practical standpoint, this intensity dependency also provides the advantage of being able to measure through optically transparent layers, allowing confocal Raman microscopes to z-scan through a material and for handheld Raman spectrometers to measure raw materials through their packaging (see Fig. 2). With more advanced techniques such as spatially offset Raman spectroscopy (SORS), one can measure through nontransparent materials, although the underlying physical process is quite different.The final consideration to be addressed is the wavelength dependencies of Raman scattering. Based upon what we have discussed so far, this may seem irrelevant, but if you dig further into the contributing factors to the Raman scattering cross-section, it can be shown that the magnitude is inversely proportional to the laser wavelength to the fourth power. As a result, the intensity of the Raman scattering is greatly increased by using shorter-wavelength lasers, but nothing comes without a cost.
The Raman effect is an extremely weak phenomena, with typical conversion efficiencies on the order of 10-7. Therefore, the signal can easily be overwhelmed by autofluorescence from the analyte. Unfortunately, many organic and biological materials have broad autofluorescence in the visible spectrum, making it difficult in many cases to use visible laser sources for these samples.
When using high-end Raman spectrometers with interchangeable lasers and diffraction gratings, it is possible to switch between different excitation wavelengths, depending on the particular analyte. Unfortunately, this is not the case for fixed-grating Raman spectrometers used in portable, low-cost instrumentation. To handle challenging analytes in this situation, near-infrared (NIR) lasers with indium gallium arsenide (InGaAs) detectors can be used to stay above fluorescence, while deep-ultraviolet (deep-UV) laser sources can stay below fluorescence. Unfortunately, both setups have drawbacks and, consequently, are only used in extreme cases.
In the case of 1064 nm excitation, the signal is very weak and the detectors tend to be noisy and expensive. Conversely, while the Raman scattering efficiency is very high in the deep-UV region, the laser sources are extremely low power and the spectral resolution (in wavenumbers) is very poor. Thus, the vast majority of fixed-grating systems have standardized on 785 nm excitation because it is the longest-wavelength laser source that still allows for fairly wide spectral range using silicon detectors.
It is also worth noting that there can be an enhancement effect if the laser excitation wavelength is at or near the peak of the UV/visible absorption band. This resonance Raman effect is sometimes powerful enough to lift the Raman signal above the fluorescence background. One particularly well-studied example is the use of blue and green lasers for the detection of antioxidant carotenoids in human skin.5
Wavelength-stabilized lasers
Armed with the understanding of how the resultant Raman spectra are intertwined with the attributes of the excitation laser, we can now look into the practical laser requirements for various Raman setups.
The first imperative is spectral stability of the laser. Typically, wavelength-stabilized lasers fall into three categories: grating-stabilized diode lasers, gas lasers, and single-frequency solid-state lasers. Even though all of these laser types are still used, the vast majority of today's Raman systems use grating-stabilized diode lasers.6 For the sake of brevity, we will examine only this type in detail.
While diodes are currently the most popular choice for Raman excitation, this was not always the case. Traditional diode lasers tend to have a high degree of uncertainty in their center wavelength because of the way in which they are grown, and as a result they are typically specified with a ±5 nm tolerance. In Raman spectroscopy, this is completely unacceptable, as precise knowledge of the starting wavelength is required to accurately measure the shift in energy of the scattered photon.While it might seem counterintuitive to use a multimode laser instead of a single-mode laser for Raman spectroscopy, the key determining factor is typically the spectral resolution of the spectrograph being used. The overall system resolution will be defined as a convolution of the laser's spectral linewidth and the spectrometer's resolution—as long as the laser linewidth is significantly less than the spectrometer's resolution, there will not be any brooding effects because of the laser. Typical multimode HECLs have a spectral linewidth from 0.15 to 0.07 nm, which is below the spectral resolution of most fixed-grating spectrometers.
Another important factor to consider when choosing which type of HECL to use in your application is the special mode profile. Single-longitudinal-mode HECLs also happen to be single spatial mode, so these lasers are ideal for demanding applications such as confocal Raman microscopy, where a diffraction-limited beam is required. Alternatively, certain applications such as surface-enhanced Raman spectroscopy (SERS) and counterfeit drug detection often have very high degrees of heterogeneity, and multi-spatial-mode lasers are often preferred.
In addition, since multimode diode lasers have a much larger gain volume, these lasers can provide up to 10X higher output power and, thus, comparable intensity at the analyte, even though they cannot be focused to as small of a spot as single-mode lasers. In both cases, the damage threshold of the sample is often lower than the achievable laser intensity, making this point moot anyway.
Understanding the complex relationship between the excitation source, analyte, collection optics, and spectrometer itself makes it easier to comprehend why it is virtually impossible to decouple the laser properties from the resultant spectral data. And although the foregoing analysis was far from exhaustive, it should provide some basic guidelines to help you choose the excitation source for your next Raman application.
REFERENCES
1. C. V. Raman et al., Nature, 121, 711 (1928).
2. C. V. Raman, Indian J. Phys., 2, 387-398 (1928).
3. E. H. Siegler, Jr. et al., Abstracts of OSU International Symposium on Molecular Spectroscopy, Y6 (1964).
4. W. Koechner, Solid-State Laser Engineering, 5, 649 (1999).
5. P. S. Benstein et al., Invest. Ophthalmol. Vis. Sci., 39, 11, 2003-2011 (1998).
6. R. V. Chimenti, Laser Focus World, 51, 11, 25 (Nov. 2015).
About the Author
Robert V. Chimenti
Director, RVC Photonics LLC
Robert V. Chimenti is the Director of RVC Photonics LLC (Pitman, NJ), as well as a Visiting Assistant Professor in the Department of Physics and Astronomy at Rowan University (Glassboro, NJ). He has earned undergraduate degrees in physics, photonics, and business administration, as well as an M.S. in Electro-Optics from the University of Dayton. Over a nearly 20-year career in optics and photonics, he has primarily focused on the development of new laser and spectroscopy applications, with a heavy emphasis on vibrational spectroscopy. He is also very heavily involved in the Federation of Analytical Chemistry and Spectroscopy Societies (FACSS), where he has served for several years as the Workshops Chair for the annual SciX conference and will be taking over as General Chair for the 2021 SciX conference.