Template technique controls crystalline structure of titanium dioxide for photonic uses
Raleigh, NC--Researchers from North Carolina State University have developed a new technique for controlling the crystalline structure of titanium dioxide at room temperature.1 The development should make titanium dioxide more efficient in a range of applications, including photovoltaic cells, hydrogen production, antimicrobial coatings, smart sensors and optical communications technologies.
“We have now developed a technique that precisely controls the phase, or crystalline structure, of titanium dioxide at room temperature—and stabilizes that phase, so it won’t change when the temperature fluctuates,” says Jay Narayan, a professor of materials science and engineering at NC State and coauthor of a paper describing the work. "This process, called phase tuning, allows us to fine-tune the structure of the titanium dioxide so that it has the optimal structure for a desired application.”
Two phases of TiO2
Titanium dioxide (TiO2) most commonly comes in one on of two major phases—rutile, or anatase. The arrangement of atoms dictates the material’s optical, chemical, and electronic properties. As a result, each phase has different characteristics. The rutile phase is better suited for use in photovoltaic cells, smart sensors, and optical communication technologies, and smart sensors. The anatase phase has characteristics that make it better suited for use as an antibacterial agent and for applications such as hydrogen production.
Traditionally, it has been a challenge to stabilize TiO2 in the desired phase, as the material tends to transform into the anatase phase below 500 °C, and into the rutile phase at temperatures above 500 °C, notes Narayan.
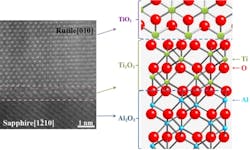
The NC State process starts with a widely available sapphire substrate that has the desired crystalline structure. The researchers then grow a template layer of titanium trioxide (Ti2O3) on the substrate. The structure of the Ti2O3 mimics the structure of the sapphire substrate. The TiO2 is then grown on top of the Ti2O3 template layer.
The structure of the TiO2 differs from the Ti2O3 -- but is dictated by the structure of that template layer. This means that one can create the TiO2 in any phase simply by modifying the structure of the Ti2O3 and sapphire substrate. This works because of a process called domain-matching epitaxy (DME). In DME, the crystalline lattice planes in the template layer line up with the lattice planes of the material being grown on that template.
The researchers have also demonstrated how this technique can be used with silicon computer-chip substrates, which can be integrated into electronics such as smart sensors.
REFERENCE:
1. M. R. Bayati et al., Applied Physics Letters, published online June 20, 2012.
About the Author
John Wallace
Senior Technical Editor (1998-2022)
John Wallace was with Laser Focus World for nearly 25 years, retiring in late June 2022. He obtained a bachelor's degree in mechanical engineering and physics at Rutgers University and a master's in optical engineering at the University of Rochester. Before becoming an editor, John worked as an engineer at RCA, Exxon, Eastman Kodak, and GCA Corporation.