New structural and optical properties of glass are identified
There is still a lot that is unknown about glass, such as how certain glasses form and how they achieve certain properties. Better understanding of this could lead to technological improvements such as scratch-free coatings and glass with unusual mechanical properties.
Over the past few years, researchers at the University of Pennsylvania (Philadelphia, PA) have been looking at properties of stable glasses, which are closely packed forms of glasses produced by depositing molecules from a vapor phase onto a cold substrate.
"There have been a lot of questions about whether this is analogous to the same amorphous state of naturally aged glasses such as amber, which are formed by just cooling a liquid and aging it for many, many years," says Zahra Fakhraai, an associate professor of chemistry at Penn.
To answer these questions, Fahkraai and Ph.D. student Tianyi Liu collaborated with chemistry professor Patrick Walsh who designed and synthesized a new molecule that has a spherical shape. According to Fakhraai, these unique molecules can never align themselves with any substrate as they are deposited. Because of this, the researchers expected the glasses to be amorphous and isotropic (that is, no overarching pattern or order).
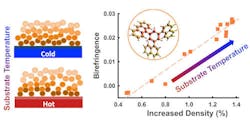
Surprisingly, the researchers noticed that these stable glasses are birefringent, which wouldn't be expected with the spherical molecules.1
In most deposited glasses, birefringence is a result of molecules aligning in a particular direction as they condensate from the vapor phase into a deep glassy state. But the birefringence patterns of the stable glasses were strange, Fakhraai said, as the researchers did not expect any orientation of the spherical molecules in the material.
After teaming up with physics professor James Kikkawa and Ph.D. student Annemarie Exarhos, who did photoluminescence experiments to look at the orientation of the molecules, and chemistry professor Joseph Subotnick and Ph.D. student Ethan Alguire, who helped with the simulations aimed at looking at the crystal structure and calculating the index of refraction of the crystal which allowed them to work out the math of the degree of birefringence or ordering in the amorphous state, the researchers confirmed their hunch that there was no orientation in the material.
Despite measuring zero order in the glass, the scientists still saw an amount of birefringence analogous to having up to 30% of the molecules perfectly ordered. Through their experiments, they found that this is due to the layer-by-layer nature of the deposition that allows molecules to pack more tightly in the direction normal to the surface during the deposition. The denser the glass, the higher the value of birefringence. This process can be controlled by changing the substrate temperature that controls the degree of densification.
Possible anisotropic mechanical properties
Since the stressors are distributed differently in and out of plane, these glasses could have different mechanical properties, which may be useful in coatings and technology. It may be possible to manipulate the orientation of a glass or its layering to give it certain properties, such as antiscratch coatings."We expect that if we were to indent the glass surface with something," Fakhraai says, "it would have different toughness versus indenting it on the side. This could change its fracture patterns or hardness or elastic properties. I think understanding how shape, orientation and packing could affect the mechanics of these coatings is one of the places where interesting applications could emerge."
According to Fakhraai, one of the most exciting pieces of this research is the fundamental aspect of now being able to show that there can be amorphous phases that are high-density. She hopes she and other researchers can apply their understanding from studying these systems to what would happen in highly aged glass.
"This tells us that we can actually make glasses that have packings that would be relevant to very well-aged glass," Fakhraai says. "This opens up the possibility of better fundamentally understanding the process by which we can make stable glasses."
This research was funded by National Science Foundation grants DMR-11-20901, DMR-1206270, CHE-1152488 and DMREF-1628407.
REFERENCE:
1. Tianyi Liu et al., Physical Review Letters (2017); doi: https://doi.org/10.1103/PhysRevLett.119.095502
About the Author
John Wallace
Senior Technical Editor (1998-2022)
John Wallace was with Laser Focus World for nearly 25 years, retiring in late June 2022. He obtained a bachelor's degree in mechanical engineering and physics at Rutgers University and a master's in optical engineering at the University of Rochester. Before becoming an editor, John worked as an engineer at RCA, Exxon, Eastman Kodak, and GCA Corporation.