NEUROSCIENCE: Small, stable quantum dots advance real-time neuronal research
The large diameters (15–35 nm) of commercial quantum dots (QDs) preclude their use in spatially confined biological environments such as cell junctions and neuronal synapses. And while small QDs (sQDs) with diameters of ~10 nm have been successfully fabricated, these sQDs typically suffer from chemical instability (stable for only hours at a time) or poor labeling specificity. Now, by using a new coating method with specially designed ligands, researchers from the University of Illinois at Urbana-Champaign, the University of Chicago, the University of Liverpool, Florida State University (Tallahassee), and the Marine Biological Laboratory (Woods Hole, MA) have developed both small and stable QDs at visible wavelengths.1
Ligand engineering
Semiconductor QDs typically have a cadmium selenide (CdSe) core with a zinc sulfide (ZnS) shell. This 2–7 nm diameter central region is then coated with water-resistant chemical and polymer layers onto which various biological markers can be attached. Unfortunately, these outer layers often result in outer diameters several tens of nanometers larger.
To improve stability and emissivity while maintaining a small diameter, the research team developed a self-assembled ligand coating that hydrophobically attaches to the nanoparticle core surface, resists desorption, and packs densely on the QD surface. Using an elevated temperature (60°C) without oxygen in the coating process drives replacement of the native hydrophobic ligands with a dense layer of the new ligands, making these sQDs, with 8 to 11 nm diameters, stable in aqueous solution for more than one month. The resultant sQDs are very fluorescent with a typical 28-percent quantum yield and can be fabricated in visible emission wavelengths from 527 to 655 nm.
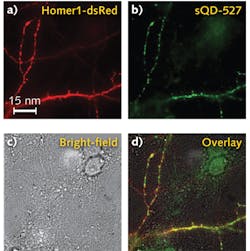
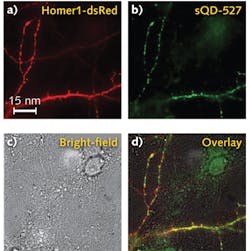
Because nonspecific interactions between sQDs and neurons or the substrate onto which neuronal tissue is placed can be a major limitation in labeling success for live-neuron applications, binding specificity for these sQDs was tested. It was discovered that high labeling specificity (meaning little nonspecific interactions with other biological elements involved in a specific application) was observed.
Neuronal research
With a clear signal, the researchers were able to tell if the sQDs or the big QDs (bQDs) were specifically getting inside neuronal synapses. They used a super-resolution microscopy technique, which won the 2014 Nobel Prize, to locate the synapses and to precisely tell where the quantum dots were. It turns out the bQDs were primarily labeling receptors that were extra-synaptic, while sQDs were primarily labeling those receptors inside the synapses.
Outside of the synapses, the receptors were highly mobile, but inside, the diffusion was much less, although non-zero. "You have to be careful about where you're looking," says professor Paul Selvin of the University of Illinois, who led the work. "Why there is some residual diffusion of the small quantum dots remains unclear."
Given the brightness, stability, and small size of these new QDs, there are plenty of pure and applied projects. "These new sQDs," says Selvin, "have tremendous potential for studying the synapse and other neurological functions, as well as other biological systems such as DNA sequencing and mapping." He cites plans to study what a memory looks like. "Take a mouse, train it to do something, and by using the small quantum dots, you can get an image of what changes in the mouse's brain."
Application for studying post-traumatic stress syndrome, or Alzheimer's syndrome, where memories are present, or are forgotten, is a possibility, he says. More immediately, Selvin has plans to use the quantum dots for measuring DNA mapping. "To get the number of genes is something that DNA sequencing has trouble doing, but we can potentially do it by making a road map of the DNA with our bright quantum dots," he notes.
1. E. Cai et al., Angewandte Chemie online, September 26, 2014; doi:10.1002/anie.201405735.