Biophotonics techniques capture cell that can regenerate an entire organism
Researchers at the Stowers Institute for Medical Research (Kansas City, MO) have developed a biophotonics technique that combines genomics, single-cell analysis, flow cytometry, and imaging to capture and isolate the cell capable of regenerating an entire organism. The findings will likely propel biological studies on highly regenerative organisms like planarians and also inform regenerative medicine efforts for other organisms like humans that have less regenerative capacity.
Related: Marriage of microscopy techniques reveals 3D structure of critical protein complex
"Our finding essentially says that this is no longer an abstraction, that there truly is a cellular entity that can restore regenerative capacities to animals that have lost it and that such entity can now be purified alive and studied in detail," says Alejandro Sánchez Alvarado, Ph.D., an investigator at the Stowers Institute and Howard Hughes Medical Institute and senior author of the study.
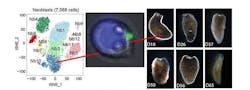
Sánchez Alvarado and his team began searching for a distinguishing characteristic that could identify this elusive cell ahead of time. One feature that had long been used to distinguish neoblasts from other cells is a stem cell marker known as piwi-1, so postdoctoral research associate An Zeng, Ph.D., decided to start there. First, he separated the cells that expressed this marker from those that did not. Then, he noticed the cells could be separated into two groups – one that expressed high levels of piwi (aptly called piwi-high) and another that expressed low levels of piwi (called piwi-low). When Zeng studied the members of these two groups, he found only those that were piwi-high fit the molecular definition of neoblasts.
Next, Zeng selected 8000 or so of the piwi-high cells and analyzed their gene expression patterns. To his surprise, the cells fell not into just one or two, but 12 different subgroups. Through a process of elimination, Zeng excluded any subgroups with genetic signatures indicating that the cells were destined for a particular fate, like muscle or skin. That left him with two subgroups that could still be pluripotent, which he named Nb1 and Nb2.
The cells in subgroup Nb2 expressed a gene coding for a member of the tetraspanin protein family, a group of evolutionarily ancient and poorly understood proteins that sit on the surface of cells. Zeng made an antibody that could latch onto this protein, pulling the cells that carried it out of a mixture of other suspected neoblasts. He then transplanted the single purified cell into a planarian that had been subjected to lethal levels of radiation. Not only did these cells repopulate and rescue the irradiated animals, but they did so 14X more consistently than cells purified by older methods.
"The fact that the marker we discovered is expressed not only in planarians, but also in humans suggests that there are some conserved mechanisms that we can exploit," Sánchez Alvarado says. "I expect those first principles will be broadly applicable to any organism that ever relied on stem cells to become what they are today. And that essentially is everybody."
Full details of the work appear in the journal Cell.