Red-light-responsive protein could someday have utility in optogenetics
A team of researchers at the Graz University of Technology (TU Graz) and the Medical University of Graz (both in Graz, Austria) has functionally characterized the three-dimensional (3D) interaction between red-light receptors and enzymatic effectors. The work has implications for optogenetics (controlling genetically modified cells using light), as it could lead to future development of novel red-light regulated optogenetic tools for targeted cell stimulation.
Related: Glowing protein could serve as light source for optogenetics
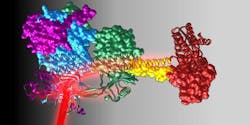
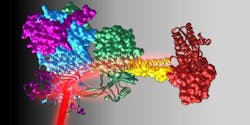
The researchers were able to observe molecular principles of sensor-effector coupling in the full-length structure of a red-light responsive protein, including detailed mechanisms of signal transmission over long distances at a molecular level. The architecture and composition of the linker element connecting the sensor and effector is very important in light regulation.
"By using a combination of x-ray structural analysis and hydrogen-deuterium exchange, by which the structural dynamics and conformational changes can be analyzed, we managed to better understand the functional characteristics of this helical coupling element," explains TU Graz biochemist Andreas Winkler, who led the work. "We were able to show that illuminating the sensor with red light resulted in a rotation-like change in the coiled coil linker region, which in turn effects the enzymatic activity of the neighboring effector." In this way, the researchers were able to determine structural details of a red-light regulated full-length system and describe molecular mechanisms of signal transduction.
The research helps to clarify the modularity of naturally occurring protein domains, which could enable development of new optogenetic tools. Diverse combinations of different sensor modules are found in nature, such as red-light sensors, blue-light sensors, and pH sensors—sometimes with identical and sometimes different effectors. From this, the researchers conclude that there are molecular similarities in signal transduction and, therefore, that rational and completely arbitrary combinations of sensors and effectors that do not occur in nature are conceivable.
"We are currently limited to naturally occurring systems to a great extent in the use of directly regulated enzymatic functionalities," Winkler says. "The long-term aim is to generate new light-regulated systems which can overcome the limitations of nature and which would be of great interest for different applications in optogenetics."
Full details of the work appear in the journal Science Advances; for more information, please visit http://advances.sciencemag.org/content/3/3/e1602498.