New utility for optogenetics could rule out potentially dangerous drugs faster
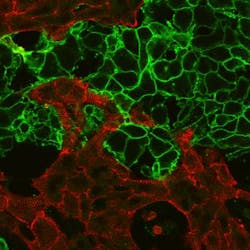
A team of researchers at George Washington University (GW; Washington, DC) has developed a faster method to predict whether potential new drugs will cause heart arrhythmias using optogenetics, a technique that uses light to control cells. While optogenetics has been used in neuroscience for a decade, this technique is relatively new in cardiac research.
Related: Optogenetics identifies receptor linked to cancer, expanding into drug discovery
In the newly developed system, the research team used light to both make the cardiac cells beat and to optically measure their response. This allowed them to automate the drug-testing process, offering a fast, new way to rule out potentially dangerous drugs. The technique streamlines a primarily manual process that researchers carry out to comply with FDA testing requirements and ensure the safety of the drugs. The team's current system can test 30,000 light-responsive cells in fewer than 10 minutes—a far shorter timeline than the standard practice, which can take hours or even years.
Traditionally, the most reliable method for drug testing is to measure their response manually by directly sticking probes into the cell, and high-throughput technologies would speed up this practice. The research team's approach, which is high-throughput and works with actual cardiac cells, helps provide a more complete picture of how the drug could affect the heart early in the drug development process, saving time and resources that can be spent on other new drug candidates.
"The benefit of optical stimulation and optical recording is that it provides a way to dynamically control millions of cells simultaneously without needing to come into contact with the sample," says Aleks Klimas, first author on the paper and a PhD student of Emilia Entcheva, professor of biomedical engineering at GW and senior author of the paper. "This not only allows you to perform faster testing, but also provides a safer way to do measurements if you're testing hazardous materials."
Separate from its uses in drug discovery, the method can also be used by scientists to confirm that stem cells have properly matured into heart cells. The new technique will help them improve the quality of these newly created heart cells and speed up their deployment in the clinic.
The technology has the potential to be adopted by pharmaceutical companies to expedite the drug development process. Following the completion of her PhD in biomedical engineering at GW, Klimas hopes to commercialize the idea and develop a business to market the device.
Full details of the work appear in the journal Nature Communications; for more information, please visit http://dx.doi.org/10.1038/ncomms11542.