BioOptics Breakthroughs
Two-photon microscopy enables view of brain’s first interpretations
An in vivo two-photon laser-scanning microscope has enabled observation of a phenomenon never seen before. The system enabled Duke University (Durham, NC) scientists to see inside the brain of a baby ferret as it opened its eyes for the first time and learned to interpret moving images. “This is the first time that anyone has been able to watch as visual experience selectively shapes the functional properties of individual neurons,” said David Fitzpatrick, professor of neurobiology and director of the Duke Institute for Brain Sciences. “These results emphasize just how important experience is for the early development of brain circuits.” The group’s findings were published online in Nature.
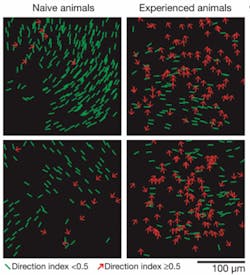
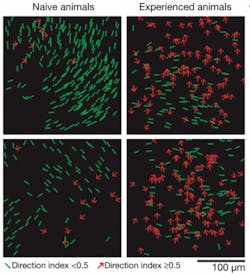
As the ferret learned to discriminate one pattern of motion from another over several hours, the researchers could see large numbers of individual neurons in the visual cortex develop specific responses and become organized into cortical columns. Additional experiments confirmed that the changes were dependent on the neurons being activated by the animal’s experience with moving visual images.
The laser microscope enabled viewing of a virtual slice of living tissue a few microns thick, and up to 300 µm in depth. A fluorescent dye sensitive to calcium allowed the scientists to detect changes in the activity of individual neurons.
“Before experience with a moving stimulus, individual neurons respond almost equally to opposite directions of motion and there is little order in the way they are arranged,” Fitzpatrick said. “But as a result of experience with moving images, their response to a particular direction of motion strengthens and they begin to act like their neighbors, forming columns of neurons with similar preferences. We have been able to visualize the self-organizing process by which the brain uses experience to guide the construction of circuits that are critical for interpreting moving stimuli.”
Fitzpatrick believes the team’s findings can be generalized to other brain regions and will be of value in understanding neurological and psychiatric disorders.
PDT makes molecular-level skin improvements
Photodynamic therapy (PDT) produces changes–apparently consistent with increased collagen production and improved appearance–at the molecular level in aging skin, says a report in the October issue of Archives of Dermatology. “Several visible and infrared lasers and light sources have been reported to produce various positive changes in the clinical and histologic [microscopic] appearance of the skin.”
Jeffrey S. Orringer, M.D., and colleagues at the University of Michigan Medical School (Ann Arbor, MI) studied treatment in 25 adults aged 54 to 83 with sun-damaged forearms. Before treatment, the degree of skin damage was rated and a tissue sample was taken. A solution containing 5-aminolevulinic acid (5-ALA) was applied to the treatment site and left on for three hours; the skin was then washed and treated with a pulsed-dye laser. Participants returned for reexamination and to provide more biopsy samples four to five times during the following six months.
After treatment, the tissue samples demonstrated a five-fold increase in levels of Ki67, a protein thought to play a fundamental role in the growth and development of new skin cells. The epidermis increased in thickness 1.4-fold. Levels of enzymes and other compounds associated with the production of collagen, the main structural protein in the skin, also were increased.
“Photodynamic therapy with the specific treatment regimen employed produces statistically significant quantitative cutaneous molecular changes,” the authors conclude. “Although our molecular measurements cannot yet precisely predict clinical outcomes for a single given patient, taken together they are very much in keeping with the bulk of the clinical literature.”
Nanostars golden for SERS sensitivity amplification
Scientists at the National Institute of Standards and Technology (NIST; Bethesda, MD) have demonstrated that the optical qualities of a certain type of nanoparticle–gold nanostars–makes them particularly useful for chemical and biological sensing and imaging. These uniquely shaped nanoparticles may one day be used in a range of applications from disease diagnostics to contraband identification.
The use of surface-enhanced Raman spectroscopy (SERS), which relies on metallic nanoparticles to amplify signals from molecules present only in trace quantities, enabled the discovery. Shining laser light on an aqueous solution containing the nanoparticles and the molecule of interest, scientists are able to determine characteristics of both components by monitoring scattered light. The strength of scattered light contains an identifying fingerprint for the molecule known as its vibrational signature. With nanoparticles amplifying the signature, it is possible to detect a very low concentration of molecules in a solution.
The NIST team tested the optical properties of the nanostars using two target molecules, 2-mercaptopyridine and crystal violet. These molecules were selected because of their structural similarity to biological molecules and their large number of delocalized electrons, a characteristic that lends itself to SERS. NIST researchers found that the Raman signal of 2-mercaptopyridine was 100,000 stronger when nanostars were present in the solution. The stars were also shown to be particularly capable of enhancing the signature of crystal violet, delivering a signal about 10 times stronger than the previous winner, nanorods. Both the nanostars and the nanorods outperformed the nanospheres commonly used for Raman enhancement.
NIST physicist Angela Hight Walker and her team perfected the process for making gold nanostars, building them from the bottom-up using surface alterations to manipulate their growth and control their shape. Once suspended in a solution, the team guided the nanostars to gather together to form multiple “hot spots,” where the enhancement is dramatically larger than for a single nanostar.
Hight Walker says the star-shaped particles can now be created in volume quantities. The work is published in the Sept. 24 issue of the Journal of Raman Spectroscopy.
Single-cell analysis reveals cancer growth patterns
The October issue of the journal Cancer Cell reports research indicating the benefits of single-cell analysis for understanding some complex cancers.
“We had a strong hunch that we could use ‘deranged’ cellular signaling to track how cancer-cell populations behave at diagnosis through therapy, as well as during remission or return of the cancer,” said Dr. Garry P. Nolan of the Stanford University School of Medicine (Stanford, CA), who led the research along with Dr. Mignon L. Loh of the University of California San Francisco Children’s Hospital and the Helen Diller Family Comprehensive Cancer Center. “By measuring how signaling proteins respond to certain stimuli at diagnosis and which are modified by resistant cancers, we are essentially monitoring key highways that cancers use to drive their own growth.”
The researchers add that single-cell diagnosis promises early detection and yields insights into how cancer cells are responding or adapting to therapy. “We should eventually be able to predict those pathways cancer cells might be using to circumvent current therapies and more intelligently direct the patient towards alternative treatments,” said Nolan.
Juvenile myelomonocytic leukemia (JMML), which is difficult to diagnose and has a complex molecular profile, provided focus for the research. Although genetic lesions impacting Ras signaling and alterations downstream of the activated GM-CSF receptor (both linked with inappropriate cell growth and survival) have been linked with JMML, there are very few methods for identifying therapeutic agents and assessing efficacy.
The scientists used flow cytometry to profile signaling at the single-cell level, including molecules associated with GM-CSF and Ras signaling, for the presence of primary JMML cells with altered signaling behavior that correlated with disease physiology. Cells samples came from JMML patients, healthy individuals and patients with other myeloproliferative disorders, some who had initially been diagnosed with JMML.
An unexpected STAT5 signaling signature was evident in most of the JMML patients, suggesting a critical role for JAK-STAT signaling in the biological mechanism of this cancer and suggesting potential targets for future therapies.
“This work successfully used single-cell profiling to follow patients over time and show that disease status in JMML–at diagnosis, remission, relapse and transformation–was indicated by a subset of cells with an abnormal signaling profile,” says Dr. Loh. “Revealing cell subpopulations, even rare cells, that are associated with disease opens additional avenues for measuring minimal residual disease, assessing biochemical effects of targeted therapies at the single-cell level, and understanding drug actions and mechanisms of diseases of heterogeneous origins and manifestations in diverse patient populations.”