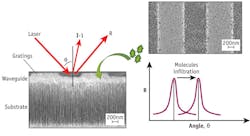
BIOSENSORS/DNA SEQUENCING: Huge sensing surface in a small space enables superior sensitivity
Originally designed to detect the presence of particular DNA sequences and thus identify whether a person is predisposed to heart disease or certain cancer types, a biosensor created by Vanderbilt University researchers promises to also enable detection of tiny environmental toxins. Current biological chemical sensors are limited because their size is often disproportionately large compared with certain target chemicals with molecules too small to register detection. According to the authors of a paper describing the innovation, it’s possible to eliminate this challenge by making sensors with features closer in size to the molecules being detected.1
A porous silicon material, which acts as a sponge and can be “seeded” with substances that change its properties, was the key to producing a detector highly sensitive to small molecules. Capturing a particular sequence of DNA involves seeding the sensor with a single strand of DNA, so that only the complementary strand can attach to it (everything else gets rinsed away).
The sensor’s enormous surface area relative to its small size is significant: a cube measuring 3 cm on each side that’s made of the porous silicon material provides a surface area nearly 10,000 times greater than the same size cube with a flat surface—and thus offers a much greater ability to capture molecules of interest.
By using a “grating,” a type of surface texturing of the porous silicon, light can be delivered in a very simple and compact way to detect change in the sensor’s properties and determine whether any molecules of interest have been captured. “When we infiltrate the molecules that we want to detect and they stay in the sensor and attach, they change the optical density of the porous silicon and, consequently, the angle or the color of light that comes back out,” says paper co-author Susan Weiss. “By knowing how much the angle changes, for example, we can quantify how many molecules are present. So not only can we identify our DNA sequence or toxin, but we can also know how much is present.”
1. X. Wei and S.M. Weiss, Opt. Exp. 19 (12), 11330–11339 (2011).
About the Author

Barbara Gefvert
Editor-in-Chief, BioOptics World (2008-2020)
Barbara G. Gefvert has been a science and technology editor and writer since 1987, and served as editor in chief on multiple publications, including Sensors magazine for nearly a decade.