PHOTOACOUSTICS: In-vivo photoacoustic scanner images tumor vasculature and therapy response
Photoacoustics is the science of converting light-induced ultrasonic emissions to images. In the case of imaging cancerous tumor vasculature, researchers at University College London (UCL; London, England) have developed a new photoacoustic scanner based on a Fabry-Perot polymer film ultrasound sensor with a unique image reconstruction algorithm that can noninvasively image tumors at depths up to 10 mm.1
The in-vivo and label-free technique improves over traditional magnetic resonance imaging (MRI) and X-ray computed tomography (CT) scans that typically require contrast agents to image blood vessels, as well as newer optical techniques such as optical coherence tomography (OCT) and multiphoton microscopy that are typically limited to soft-tissue imaging depths of 1 mm due to high scattering.
Low-scatter acoustic waves
Unlike the ballistic photons in multiphoton microscopy that suffer from high scattering in soft tissues, the near-infrared (IR) and visible light striking a tissue sample in a photoacoustic setup creates rapid heating that converts the optical energy to acoustic energy (sound waves) that scatter far less than photons. The acoustic signals, when reconstructed, reveal a picture of the absorbed energy of a tissue, which depends upon the tissue's optical properties. Acquiring images at multiple wavelengths can even reveal quantitative spectroscopic measurements of blood oxygen saturation and hemoglobin concentration, in addition to 3D depth imaging.
To study how a tumor and its surrounding blood vessels are affected by a therapeutic vascular disrupting agent, the researchers imaged, in-vivo, human colorectal adenocarcinoma xenografts in nude mice at different times using their all-optical photoacoustic scanner. In the setup, a fiber-coupled, wavelength-tunable, pulsed Nd:YAG optical parametric oscillator (OPO) laser system delivers 7 ns pulses at 50 Hz to the tissue under study. A Fabry-Perot (FP) sensor head used to detect the photoacoustic waves is placed in acoustic contact with the surface of the skin using an aqueous gel.
The FP sensor head is composed of two dichroic mirrors separated by a thin polymer spacer (40 µm) deposited onto a wedged polymer substrate to form a Fabry-Perot interferometer (FPI). The mirrors are transparent from 590–1200 nm, but highly reflective between 1500–1600 nm, allowing the excitation-laser pulses in the former wavelength range to be transmitted through the sensor head into the target tissue. Tissue absorption of the waves creates photoacoustic pulses that modulate the optical thickness of the FPI and therefore, its reflectivity. By raster-scanning a focused interrogation laser beam at 1550 nm across the surface of the FPI and recording the reflected optical power modulation at each point, a 2D map of the incident photoacoustic waves can be obtained.
From the photoacoustic signals, 3D images are formed using a time-reversal image reconstruction algorithm that compensates for the acoustic attenuation characteristic of soft tissues through a frequency-dependent amplification that exactly counteracts the tissue absorption. This significantly improves image intensity and resolution—especially at greater tissue depths.
Results
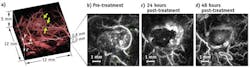
Photoacoustic images of a subcutaneous tumor treated with a therapeutic vascular disrupting agent were obtained both before and 24 and 48 hours after injection of the agent. Post-treatment images clearly show dynamic changes in the tumor vasculature due to the vascular shutdown that occurs over time, which results in strongly decreased perfusion particularly within the tumor core and thus reduced image contrast.
"The compelling in-vivo images provided by this novel photoacoustic sensing technology enable visualization of tumor-specific vascular features that cannot be observed noninvasively by existing imaging modalities to the depths reported in this study," says Paul Beard, head of the UCL photoacoustic imaging group. "This makes it a potentially powerful imaging tool for evaluating and developing new cancer treatments based on targeting tumor vasculature. Although our focus to date has been on demonstrating the technology as a preclinical research tool, there are a number of clinical skin-imaging applications that we plan to investigate, including diagnosis and treatment monitoring of skin cancers such as melanomas and characterizing abnormalities of the microcirculation associated with diabetes, venous disease, and inflammatory skin conditions such as dermatitis."
1. J. Laufer et al., J. Biomed. Opt., 17, 5 (May 2012).