LIBS leaves the lab for field work in industry and defense
Laser-induced breakdown spectroscopy (LIBS) has experienced a surge of growth in recent years, particularly for field-based applications. The development of new laser sources and portable instrumentation is helping LIBS find a niche in everything from environmental monitoring and materials analysis to homeland security, medical diagnostics, and even space-based research. In addition, the emergence of the echelle spectrometer and low-cost broadband spectrometers—both of which afford higher spectral coverage and resolution—has opened up new applications in molecular- and biological-matter detection and sorting of metals and plastics.
"If you can do it in the lab using conventional techniques, that works really well. But if you need to go out into the field and do it rapidly, that is where LIBS really shines," said David Cremers, technical staff member at Los Alamos National Laboratory (Los Alamos, NM), whose group is developing a LIBS-based instrument for terrestrial carbon sequestering and remote elemental analysis of Martian soils (see Fig. 1). "It is not going to replace the majority of lab-based instruments, but LIBS works well in industrial environments and unique situations such as forensics."
Much of the technology that goes into a typical LIBS system—laser, optical light-collection system, spectrometer, detector, and computer—has been around for years, and the technique itself has been known since the early 1960s (see "How LIBS works," p. 76). Even so, LIBS has had a tough time gaining a following in many of the industries for which it is best suited. But as the components mature and improve, and research continues to validate LIBS as an effective and even superior analytical instrument for field-based measurements of materials ranging from steel to soil to ambient air and even coffee beans, LIBS has begun to attract more funding from commercial and military investors.
"LIBS really started taking off about 1996 when people began to recognize that it is appropriate for a lot of applications in industrial process control, environmental monitoring, and space," Cremers said. "In addition, people have now developed small lasers, spectrographs, and detectors that are fairly rugged, so you can take this equipment out of the lab to do elemental analysis in the real world and tackle problems that conventional methods can't handle."
Technical advantages and advances
One reason for the ongoing interest in LIBS is that it has several inherent advantages over other analytical techniques. There is no need for sample preparation and results are obtained in near real time. In addition, LIBS is relatively easy to use and can provide information on virtually any element in any form.
"LIBS has tremendous advantages in terms of speed and lack of need for sample preparation," said Andrzej Miziolek, a researcher with the U.S. Army Research Laboratory (Aberdeen Proving Ground, MD), which is working with Ocean Optics (Dunedin, FL) to develop a backpack-based LIBS system for defense and homeland-security applications (see Fig. 2). "What really makes it unique is that LIBS can spark in so many materials, so you can do surface analysis and make depth profiles in liquids, chemicals, metals, aerosols, and bioagents. I do not know of any other sensor that has that range and versatility and sensitivity."
In recent years, several advances in technology have helped LIBS begin to expand its user base. The ongoing refinement of compact solid-state lasers, for example, has led to increasingly portable and robust LIBS systems. There has also been a push to expand LIBS into a broader spectral range and to make the technology more affordable and more accessible for real-world applications. Last year Ocean Optics introduced the LIBS 2000+, which links seven spectrometers to make one broadband, high-resolution spectrometer with a spectral range from 200 to 980 nm and 0.1-nm spectral resolution—all for under $50,000. This fall the company plans to release the LIBS 3000, which is one-third the size of the LIBS 2000+ and about one-seventh the weight.
Other R&D efforts are targeting multi-element analysis. Previous generations of LIBS focused on one element at a time, using an averaging mode that required 50 to 100 shots to acquire the information necessary to identify a single element. But here again, technical advances—particularly in computing power—have paved the way for single-shot particle analysis of multiple elements.
"Just three years ago you couldn't do this," Miziolek said. "But now we are in a position that we can track all the key elements per laser shot and keep track of that information."
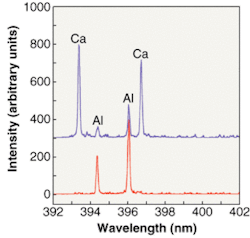
This capability is especially important for analyzing more-complex environments such as ambient air. At the University of Florida (Gainesville), for example, David Hahn and his group have been using LIBS to look at individual particles in aerosols and other air-based environments and provide quantitative, not just qualitative, information about their composition, which should prove important for environmental monitoring (see Fig. 3). So far Hahn's group has used LIBS to measure ambient air, smokestack emissions, arsenic in pressure-treated lumber, and the rise and fall of magnesium in the air from fireworks set off on the Fourth of July.
"People are very interested in fine particulate matter (less than 2.5 µm) because there is a worldwide consensus that this matter is very much a health issue," Hahn said. "And LIBS is a very good potential candidate to measure this because it can do both composition and size, providing real-time in situ analysis of individuals particles."
The military is also interested in single-element analysis of bioaerosols, particularly for security applications such as anthrax detection and identifying airborne infectious diseases. A recent study by John Hybl at MIT Lincoln Laboratory (Lexington, MA) sponsored by the U.S. Army demonstrated the use of LIBS for characterizing various bioaerosols, including bacillus globigii, viruses, fungal spores, and pollen. The study concluded that combining single-particle LIBS with fluorescence-sensing technology should lead to a high-discrimination bioaerosol classifier with the potential to replace slower, more-expensive antibody, culture, or DNA-based techniques.
Breaking new ground
Another advantage of LIBS is that it lends itself to remote materials analysis, making it ideal for operating in hazardous industrial conditions. The technique has been used in Europe for several years by the steel industry for process monitoring and quality control of molten materials. For example, under the direction of Javier Laserna, researchers at the University of Malaga (Spain) have developed a remote LIBS-based system capable of analyzing targets up to 50 m away. In this instance, the laser process offers an alternative to electric-spark technologies, which operate in a similar fashion but must come in contact with the material in order to do compositional analysis, making them impractical for fast-moving or molten materials.
"I believe LIBS will have the biggest impact, in terms of commercial viability, in industrial applications in which compositional analysis of hazardous, high-temperature, and/or fast-moving materials is required, such as on-line process control of liquid alloys and glasses, characterization of radioactive materials, and rapid identification of materials during waste recycling," said Andrew Whitehouse, managing director at Applied Photonics (Skipton, England), which has been developing custom LIBS instruments for industrial measurement applications since 1998.
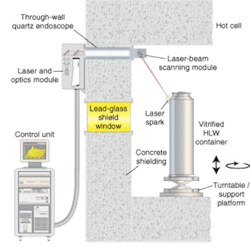
Applied Photonics has taken this same approach into the nuclear industry (see Fig. 4). Here again, the remote capabilities of LIBS, either in conjunction with an optical endoscope or a fiberoptic cable, makes it possible to perform in situ materials analysis. In situ measurements are important to the nuclear industry because they obviate the need to take physical samples of the materials for off-line laboratory analysis—a time-consuming and expensive operation requiring specialized techniques and facilities. Potential uses of LIBS include nuclear decommissioning and radioactive waste processing in which even basic identification of a material in a highly radioactive environment is very difficult by conventional means.
Other emerging LIBS applications include food analysis and archaeology. Researchers from ARL used LIBS to analyze the elemental content of coffee beans and the soil in which the beans were grown; the goal was to differentiate the country of origin of the beans by correlating the LIBS spectrum with the mineral content of the soils. The U.S. Food and Drug Administration is interested in ensuring that products imported into the United States come from the counties they are supposed to be from, which is important for both political and health reasons.
In archaeology, researchers have known for some time that LIBS offers a safe, inexpensive, noninvasive technique for analyzing the elemental content of old paintings and other artifacts. A recent collaboration between the Institute for Aegean Prehistory (Philadelphia, PA) and the Foundation for Research and Technology-Hellas (Crete, Greece) has yielded a prototype compact LIBS system that can detect paint composition, which in turn can help determine where an object came from and exactly how old it is.
While LIBS has clearly shown itself to be a viable analytical tool for a broad spectrum of field-based applications, further work remains. In particular, there is much interest in achieving more quantitative analysis using LIBS, along with a better ability to interpret the data that is gathered. Another issue is standardization of LIBS methodologies, calibration routines, and the process itself.
"The microanalytical and transient nature of LIBS makes it difficult to obtain high-precision quantitative measurements of minor elements," Whitehouse said. "So trying to standardize how we perform calibration is important, but we also need to better understand the laser-material interaction and the dynamics of the laser-produced plasma."
REFERENCE
- K. Bell, Laser Focus World 37, 8, S7 (August 2001).
How LIBS works
Laser-induced breakdown spectroscopy (also known as laser-spark spectroscopy or laser-induced plasma spectrometry) is a simple yet powerful tool that uses atomic emission to nondestructively detect and measure virtually any element in a solid, liquid, or aerosol.1 A laser beam, most often from a pulsed (50 to 100 mJ) 1064- or 532-nm Nd:YAG laser, is focused onto the sample material, generating a plasma or spark at the surface (see figure). Because the temperature in the plasma is typically around 10,000 K, the species present are turned into electronically excited atoms and ions.
As the excited species decay, they emit radiation, typically in the ultraviolet to near-infrared range, in a characteristic spectrum. Light from the spark is gathered and analyzed using a spectrophotometer. The signal light is weak in comparison to the plasma-inducing laser pulse, so a highly sensitive, gated detector is used with a filter to eliminate the laser light. The detector is typically either a charge-coupled-device array, a camera, or, in rare cases, a photomultiplier. Intensified detectors are a common choice. Cameras and arrays can give information on a range of wavelengths at once, relying on the known angle-wavelength relationship of the spectrometer.