CRDS measures atmospheric CO2
BRUCE RICHMAN, CHRIS RELLA, ERIC CROSSON, and BARBARA PALDUS
Are we transforming Earth into a big greenhouse? During the 20th century, global mean surface temperature increased by 0.6°C, global mean sea level increased at an average annual rate of 1 to 2 mm per year, and nonpolar glaciers retreated significantly. And yet, though the scientific evidence of climate changes is clear and nearly unquestioned, the degree to which human activities have affected the generation of greenhouse gases and the effect these gases have on global change remain questions of fierce political debate.
The international scientific community has proposed addressing these questions by creating a worldwide network of sensors to gather spatially and temporally resolved greenhouse-gas data that will lead to more-precise atmospheric models. To date, the most actively studied greenhouse gases are carbon dioxide (CO2), carbon monoxide (CO), and methane (CH4). Climatic variations in the concentration of these gases are small fractional changes, typically less than 1% of the concentration. For example, the atmospheric concentration of CO2 has increased by about 31%, from 280 parts per million (ppm) before the year 1750, to 368 ppm in the year 2000. Thus, sensors used in an international monitoring network must have very high accuracy, excellent precision, and behave uniformly across the globe.
Instrument requirements
New instruments used in climatic research must meet stringent performance requirements. These include precision, which is the repeatability of a measurement, defined by the standard deviation of a set of measurements on a single sample; accuracy, which is the ability to determine the exact concentration when measuring a gold standard (and which is defined by the average of the difference between the measured concentration and the gold standard concentration of the sample); linearity, which is the ability to match measurements to a set of gold standard references with a slope of 1 and an intercept of 0.000; memory, which is the ability to purge the previous sample adequately so that a measurement outcome is not affected by the sample history; and absolute zero, which is the ability to detect zero concentration of the target species (that is, when the species is absent, the measurement should produce a zero reading).
Today, instruments based on nondispersive IR (NDIR) measure concentrations by comparing sample absorption to that of a reference gas from a bottle. In turn, the bottle is itself calibrated to a gold standard. These NDIR methods are temperature-dependent and have a tendency to drift, so that frequent field calibration is required. Moreover, the calibration procedures and materials themselves can easily introduce drift, so that errors accumulate and produce unacceptable discrepancies over time.
Traditional absorption spectroscopy
Absorption spectroscopy measures the difference in intensity between the incident light intensity, I0, and the transmitted light intensity, I(x,λ). Beer’s law relates the absorbed light to the sample absorption, α(λ), as:
I(x,λ) = I0 e−α(λ)x (1)
where λ is wavelength and x is path length. Absorption is related to concentration, C, through the extinction coefficient, ε(λ)namely α(λ) = Cε(λ). The extinction coefficient is measured from the slope of a calibration curve relating concentration to absorption.
Equation 1 implies that the detection limit depends on the path length through the sample and on the intensity noise of the light source and detector. We define the minimum detectable absorption loss (MDAL) as the normalized standard deviation of the smallest detectable change in absorption (MDAL typically has units of cm−1). Sensitivity is defined as the achievable MDAL in a one-second measurement interval, and has units of cm−1 Hz−1/2. Sensitivity is a figure of merit for any absorption-based technique.
Although many species can absorb light at one or more wavelengths, the total spectral profile of any particular species remains unique. The ability of a spectrometer to distinguish between two different species absorbing at similar wavelengths is called selectivity. The instrument must be able to resolve different absorption peaks. Thus, selectivity depends on spectral resolution. Spectral resolution is another figure of merit for a spectrometer.
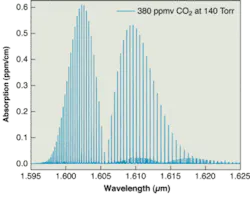
A near-IR absorption spectrum of CO2 illustrates that good precision is a direct consequence of high spectral resolution and sensitivity (see Fig. 1). The peak height, and therefore accuracy, depends on sample temperature and pressure control. Good linearity is achieved when both precision and accuracy remain high over the operating dynamic range. Memory depends on the sample-handling system and cell volume. It is desirable to minimize dead-zone volumes in the instrument. Absolute zero requires that the instrument provide an absolute measurement of the optical loss, so that a zero measurement is independent of drifts in the subcomponents or changes in the operating environment.
Cavity-ring-down spectroscopy of CO2
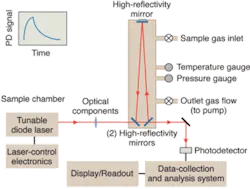
Cavity-ring-down spectroscopy (CRDS) uses a stable optical resonator called the ring-down cavity (RDC). The technique is based on the principle of measuring the rate of exponential decay of light intensity inside the RDC.1 Once sufficient light is injected into the RDC from a laser source, the input light is interrupted, and the decay of the stored light transmitted out of the cavity through one of the RDC mirrors is monitored using a photodetector (see Fig. 2).For a given wavelength, the decay rate is proportional to the total optical losses inside the RDC, which comprise the round-trip scattering and mirror-transmission losses, and sample absorbance. For typical RDC mirrors (99.995%), the effective path length can exceed 20,000 times the round-trip length of the RDC. An MDAL of 5 × 10−10 cm−1 can easily be achieved. The highest sensitivity achieved using CRDS is 1 × 10−12 cm−1 Hz−1/2. Moreover, a stable optical RDC can accomplish this sensitivity enhancement for sample volumes as small as 25 mL. The benefits of smaller sample volumes are manifested in faster flow rates through the system and reduced sample memory.
The sample concentration can be found from the spectrum obtained by taking the difference in decay constant between the baseline (concentration of zero) and the absorption peak. If the absorption cross-section and line-shape parameters of the sample are known, then the concentration of the sample can be computed. The CRDS measurement is not dependent on either the light intensity inside the cavity or on the physical sample path length; CRDS provides an absolute measurement of optical loss, from which highly accurate concentration measurements can be obtained without frequent recalibration.
Putting CRDS to the test
At Picarro, CRDS has been implemented using a triangular ring cavity and directly modulated diode-laser sources, both patented. The single-shot MDAL was 6 × 10−10 cm−1. The instrument achieved a spectral resolution having a standard deviation of 3 MHz, including noise in measuring and setting the laser wavelength. A thermal-control system maintained the RDC temperature (see Fig. 3).When nitrogen or nitrogen saturated in water vapor was flowed through the instrument, the concentration reading was zero, indicating no interferences from water. No zero drift was observed when the external temperature was varied. No additional calibration was required during operation over several months of testing.
Analyzers based on CRDS have the potential to offer better measurements of climatic gases. Such instruments could also be applied to numerous other applications, such as bioremediation, greenhouse monitoring, entomological studies, plant-physiology research, and oceanography.
Acknowledgments
The authors would like to acknowledge the contributions of Bernard Fidric, Steven Sanders, Alex Kachanov, Sze Tan, and Edward Wahl to this research. We would like to thank the Department of Energy (DOE), which has made this work possible through its SBIR grant programs.
REFERENCE
1. K. W. Busch, M. A. Busch, Cavity Ringdown Spectroscopy: An Ultratrace Absorption Measurement Technique, ACS Symposium Series 720, Oxford, 1997.
Bruce Richman is laser R&D manager, Chris Rella is director of optical design, Eric Crosson is vice president of R&D, and Barbara Paldus is chief technical officer and vice president of engineering at Picarro, 480 Oakmead Parkway, Sunnyvale, CA 94085; e-mail: [email protected].