Optogenetic brain stimulation reduces alcohol consumption in rats
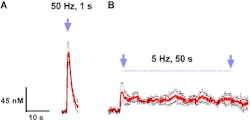
Winston-Salem, NC--Researchers at Wake Forest University and Wake Forest School of Medicine (both in Winston-Salem, NC) working with researchers from SUNY University at Buffalo (Buffalo, NY) and the University of North Carolina at Chapel Hill have demonstrated thatlow-frequency (5Hz) optical activation of dopamine neurons significantly delays the first drink and reduces a drinking binge by 54% for rats undergoing targeted optogenetic stimulation.
Alcohol consumption has been shown to be implicated in more than 60 human medical conditions, including malignant cancers, neuropsychiatric and cardiovascular disorders, gastrointestinal disease, fetal alcohol syndrome, and intentional and unintentional injuries. Despite the prevalence of alcohol consumption and abuse, the underlying neurochemical alterations that play a primary role in developing and maintaining such abuse are not known.
Over the last two decades, research has pointed to dopamine (DA) as the primary neurotransmitter that is involved in positive reward and pleasurable conditions. The VTA (ventral tegmental region) is an important region of the dopamine system that is connected by dopaminergic neurons to another brain area, the nucleus accumbens (NA). This area plays an integral role in the regulation of motivated behaviors, and alterations of this circuit have long been hypothesized to contribute to the cause of alcoholism. However, until recently technical limitations have made it impossible to investigate the causal role of DA transmission in mediating these behaviors. Using newly developed optogenetic tools, the researchers explored the causal relationship between specific patterns of VTA dopamine cell activation using light and abusive drinking behaviors in rats.
Many studies have implicated a DA mechanism in the regulation of ethanol drinking. However, it is not possible to selectively stimulate DA release from discrete DA neurons using electrical or chemical stimulation. This is absolutely necessary to test the causal link between DA neuron activation and drinking. Optogenetics allowed them to optically stimulate dopaminergic cells with exceptional selectivity and temporal precision.
While several transgenic strategies have been developed to target opsins (light-sensitive proteins) to dopaminergic neurons in rodents, the research team developed a novel viral approach that can be used in any mammalian species and potentially even humans. Here, the expression of channelrhodopsin-2 (ChR2), a mutated cell membrane protein that opens ion channels in response to light, was driven by a tyrosine hydroxylase (TH) promoter. The TH promoter confined production of ChR2 to only dopamine neurons, and not any other type of neurons; laser light was then applied to the VTA region to stimulate DA transmission.
The research team showed that they could deliver the ChR2 gene to dopamine neurons, allowing them to use the light to selectively control the timing/amount of DA released in the NA without affecting DA dynamics in the neighboring area (in the bordering brain region). They also showed that they could use optogenetic activation of the VTA to reduce the consumption of ethanol in rats. Finally, the team demonstrated that only specific optogenetic light pulses (frequency and duration) succeeded in affecting drinking behavior.
SOURCE: SPIE Biomedical Optics & Medical Imaging; http://spie.org/x106290.xml?WT.mc_id=KNRBIOMEDE
About the Author

Gail Overton
Senior Editor (2004-2020)
Gail has more than 30 years of engineering, marketing, product management, and editorial experience in the photonics and optical communications industry. Before joining the staff at Laser Focus World in 2004, she held many product management and product marketing roles in the fiber-optics industry, most notably at Hughes (El Segundo, CA), GTE Labs (Waltham, MA), Corning (Corning, NY), Photon Kinetics (Beaverton, OR), and Newport Corporation (Irvine, CA). During her marketing career, Gail published articles in WDM Solutions and Sensors magazine and traveled internationally to conduct product and sales training. Gail received her BS degree in physics, with an emphasis in optics, from San Diego State University in San Diego, CA in May 1986.