Simultaneous imaging benefits biology
Scientists typically use the various three-dimensional optical-imaging techniques independently when taking measurements. Even an experiment involving more than one two-photon technique would include two separate single-channel measurements taken at different times. Now researchers at the University of Sydney (Sydney, Australia) have demonstrated the benefits of imaging biological specimens—specifically DNA and chromosomes—using two or more imaging modes simultaneously. In one experiment, the group demonstrated simultaneous high-resolution two-photon excited fluorescence (TPF) and second-harmonic-generation (SHG) imaging of fruit-fly polytene chromosomes using a femtosecond laser and a modified off-the-shelf confocal microscope (see figure). The two interaction mechanisms produced by these techniques provide complementary information on the structure and nonlinear properties of the specimens beyond that possible with either imaging process alone.
Nonlinear optical microscopy techniques such as TPF and SHG—which are based on the interaction between the high peak-power density that exists at the laser focus and the nonlinear optical properties of the specimen in the focal region—produce spatial variation in this region that leads to a soft-aperture effect. This produces optical sectioning capability without the need for a confocal pinhole. According to the Sydney research team, such techniques allow greater imaging depth when using longer wavelengths, as well as improved optical detection (higher sensitivity is beneficial with unstained specimens) and relatively high interaction efficiency, depending on the techniques used.
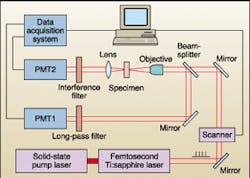
For both the DNA and chromosome imaging experiments, the Australian scientists used a modified inverted Leica confocal microscope coupled to a Coherent Mira 900 femtosecond Ti:sapphire laser with an output range from 720 to 860 nm and a pulse width of 150 fs. This pulse width allowed the group to set up the system without using an adjustable group-velocity-dispersion precompensation arrangement.
The beam-scanning microscope provided an acquisition rate of up to four 512 x 512-pixel images per second using a X40/0.75NA air objective, with the system operating simultaneously in both transmission and reflection modes (for SHG and fluorescence, respectively). For the fluorescence channel, the scientists used a broadband dichroic. The fluorescence-detection bandwidth was determined by a long-pass filter with a cutoff wavelength approaching 450 nm. To produce the SHG channel, the group excluded the 800-nm fundamental beam from the detector using a 400-nm interference filter with a bandwidth of 20 nm full-width at half-maximum and a rejection ratio at 800 nm exceeding 105. Resolution for both modes was approximately 0.5 µm.
The research team believes that simultaneous techniques such as SHG and TPF can be beneficial for nonlinear microscopic applications in which the laser pulse can interact with several nonlinear optical properties of the specimen. The resulting optical signals, while detected separately, could be combined to form enhanced, composite nonlinear optical images of the specimen. If sensitivity is not an issue, the simultaneous TPF and SHG imaging discussed here could provide higher resolution with the use of a confocal pinhole. It also will be possible to expand applications through the use of multiple detectors and an unsharp masking technique.
About the Author
Paula Noaker Powell
Senior Editor, Laser Focus World
Paula Noaker Powell was a senior editor for Laser Focus World.