Ultrafast lasers link diagnostics and therapy at the cellular level
From nanosurgery to cellular manipulation, laser-tissue interaction studies are paving the way for a new generation of medical applications.
For many years, lasers in the medical field were seen primarily as surgical tools for the ablation, cutting, or coagulation of tissue. But in the last decade some of the most successful medical-laser applications have been noninvasive (although “noninvasive” is actually a misnomer here because the effectiveness of each of these procedures requires that the laser energy enter the tissue to effect a change or achieve a biological response)-selective laser trabeculopasty, skin rejuvenation, hair removal, photodynamic therapy, low-level laser therapy, and, more recently, laser treatments of psoriasis, acne, cellulite, and dental bacteria. In addition, laser-based diagnostic tools such as optical coherence tomography and multiphoton microscopy have gained acceptance within the medical community for noninvasive imaging, while optical sensors are finding a place in the detection and monitoring of chronic and other diseases.
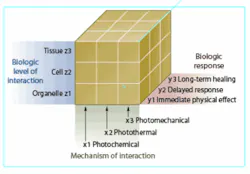
But as each of these technologies and applications has gained regulatory, physician, and consumer acceptance, researchers have continued to work to quantify the underlying mechanisms that enable them-that is, the photothermal, photochemical, and photomechanical changes that take place within the molecular structure of tissue when it is exposed to laser energy (see Fig. 1).1 In recent years, this research has led to a greater understanding of how laser-based procedures such as skin rejuvenation actually work, and has created new opportunities for analyzing and manipulating cells and cellular processes.
“Light is a great tool because it can be focused down to the submicron scale and it can penetrate into a cell without damaging the membrane,” said Eric Mazur, professor of applied physics and physics, Harvard University (Cambridge, MA). “And now we are using the light to image and target as well as to micromanipulate. Most of my setups now incorporate imaging and nanosurgery at the same time because you need the imaging to help you see what you are doing when you are micromanipulating. This is a very exciting area because it adds a new tool to the arsenal that the biochemist, biophysicist, and biologist have at their disposal to study various processes at the bioscale.”
For the last 20 years, Mazur’s research group at Harvard has been studying the dynamics of molecules, chemical reactions, and condensed matter on very short timescales (down to femtoseconds) using laser pulses. Among other things this work has led to subcellular surgery and nanosurgery, in which femtosecond laser pulses are used to selectively disrupt submicrometer-size organelles within living cells or tissue without affecting the surrounding material or compromising viability of the cell or organism. Using only a few nanojoules of laser pulse energy, Mazur and his colleagues have been able to selectively disrupt individual mitochondria in live bovine capillary epithelial cells and cleave single actin fibers in the cell cytoskeleton network of fixed human fibroblast cells (see Fig. 2).
Adela Ben-Yakar, a former member of Robert Beyer’s research group at Stanford University, visiting researcher with Mazur’s research group, and now assistant professor of mechanical engineering at the University of Texas-Austin, is also working with femtosecond lasers for potential nanosurgery applications. In research reported last December in Nature, Ben-Yakar et al. used this “laser scalpel” to snip apart a single neuron in the roundworm C. elegans (commonly used in biological research because of its simple cellular structure) to study nerve regeneration without causing any damage to surrounding tissue, allowing the study of the function of individual neurons with a precision not previously possible.2
- FIGURE 2. When a femtosecond laser pulse is tightly focused into a nearly transparent biological material, energy is deposited by nonlinear absorption only in the focus where laser intensity is high, resulting in disruption of the structure in the focal volume. Researchers at Harvard University have used this technique to manipulate subcellular structures; for example, in a live endothelial cell with fluorescently tagged mitochondria (inset), femtosecond laser pulses removed one of three very closely spaced mitochondria (arrow) without affecting nearby cells.
“The human body has 150 trillion cells (vs. 1300 cells in the C. elegans), so manipulating them at the subcellular level is probably not going to have a major effect on people,” he said. “But understanding what happens at the cellular level is going to contribute to a better understanding of the body as a whole. So while I think that most of the benefit of laser nanosurgery is going to be as a research tool, it will indirectly have an effect on therapeutic medicine by providing more knowledge to enable us to develop new methods to fight and cure and prevent disease.”
Nanoparticles for nanosurgery
Another pioneer in the field of cellular nanosurgery is Alfred Vogel, senior scientist at the Institute of Biomedical Optics of the University of Lübeck (Lübeck, Germany). His investigations into photodisruption and pulsed laser ablation (including energy deposition/transport, phase transitions, ablation plume, stress confinement, bubble formation, and plasma-mediated ablation) have led to a much greater understanding of fundamental laser-tissue interactions and enabled some of today’s most well-established medical-laser applications, such as intraocular surgery.3
“When short laser pulses are focused into transparent tissue (such as the eye), you can cut inside the eye without surgically opening the eye, which makes it effective for post-cataract surgery,” Vogel said. “Without this technique, modern cataract surgery would not be possible and intraocular lenses could not be implanted the way they are routinely today.”
The focus of Vogel’s research these days is on the use of pulsed lasers for targeting cells and proteins inside the cells via plasma formation. When the beam from an ultrafast laser is focused through a microscope, the high intensity of the pulse itself triggers the formation of nonlinear plasma, which can be used to target almost anything in a cell (see Fig. 3). In addition, Vogel is collaborating with Gereon Hüttmann, also with the Institute of Biomedical Optics at the University of Lübeck, on the development of gold nanoparticles for intelligent targeting and selective manipulation of microscopic biological structures (similar research has been done by Jennifer West and colleagues at Rice University and M.D. Anderson Cancer Center, Houston, TX4). Hüttmann’s research involves coupling inorganic absorber particles to the target (via covalent bonds or antibodies) that act as well-confined heat sources after irradiation with pico- or nanosecond laser pulses. According to Hüttmann, laser-induced nanoparticle-mediated inactivation may represent a new method to selectively interfere with molecular and cellular systems because the resulting damage mechanism, which is based on physical parameters, can be varied more precisely than chemical methods by choosing appropriate particle size, pulse width, and pulse energy.
“We are working as colleagues on cellular nanosurgery with these complementary approaches,” Vogel said. “My approach requires femtosecond or picosecond pulses from a Ti:sapphire, Nd:glass, or ytterbium laser. Hüttmann also uses picosecond or shorter pulses because the pulses have to be short enough that heat production doesn’t spread too far away from the nanoparticles. And these nanoparticles are very good absorbers; if the laser pulse is short enough, just the selected protein (or even the protein membrane) is heated, denatured, and destroyed.”
The potential for this research includes being able to eliminate certain types of cells, even specific cancer cells, although the process needs to be further refined to allow the nanoparticles to be introduced directly into the cell or onto the cell surface. But Vogel says it could prove to be an effective early-stage cancer treatment that is both patient- and cancer-specific.
“Intracellular surgery is basically used for functional research to damage something in the cell and then determine its function,” he said. “But it can also be used to dissect individual cells out of the tissue compound for proteomic and genomic analysis on the cellular basis. The ideal treatment for cancer would be patient- and cancer-specific, which requires the ability to look for tumor markers (specific proteins and gene patterns) in the tumor cells. To do precise quantitative analysis of the tumor markers, you must be able to isolate the tumor cells. So if one would manage to do a very precise analysis of tumor markers using a pulsed laser with an inverted microscope, that becomes a very important adjunct to clinical therapy.”
A laser technique developed by scientists at the University of St. Andrews (St. Andrews, Scotland) could provide Vogel and his colleagues with a way to introduce their nanoparticles into human cells.5 The process, which has seen genes delivered directly into mammal cells using a laser, first involves suspending the cell to be implanted in a solution of the gene material to be introduced. A violet diode laser is then focused on the cell membrane for a matter of milliseconds, which causes the membrane to open. Researchers believe that the adaptability of the technique means it could have wide applications, including delivery of anticancer agents directly into cells, and in advanced studies of neuro-degenerative diseases such as Alzheimer’s and Parkinson’s.
“We believe that we have only touched the surface with this technology,” said Ben Agate, a key researcher on the project. “The method is simple and inexpensive and could have important biomedical implications. Since it also has the potential to assist in the cellular delivery of other biomolecules, we are now looking at other cell types to see how widely applicable the method proves to be.”❏
REFERENCES
1. S. L. Jacques, Lasers in General Surgery 72(3) (June 1992).
2. M. F. Yanik, H. Cinar, H. N. Cinar, A. Chisholm, Y. Jin, A. Ben-Yakar, Nature 432(7019) (Dec. 16, 2004)
3. A. Vogel, V. Venugopalan, Chem. Rev. 103 (2003).
4. L. R. Hirsch et al., PNAS 100, 13549, DOI:10.1073/pnas.2232479100, 3 (2003).
5. L. Paterson et al., Optics Express 13(2) 595 (Jan. 24, 2005).