Erbium is so cool
Laser cooling of atoms has led to some striking advances in physics research, including the first demonstration of “slow light,” in which a cloud of 50-μK sodium atoms in a degenerate quantum state slowed light to a speed of 17 m/s (see Laser Focus World, April 1999, p. 16). Laser-cooled atoms may also have practical applications in creating frequency standards and fabricating nanostructures.
Because each type of atom has unique properties, the ability to cool different types of atoms will only open up more opportunities. A group of scientists at the National Institute of Standards and Technology (NIST; Gaithersburg, MD) has now added erbium (Er) to the list.1 Erbium is especially interesting to the photonics community because it is the dopant of choice for many photonic devices that operate at telecommunications wavelengths.
When laser light is tuned to an atomic resonance, the atoms absorb photons from the laser and rescatter them into all directions, essentially absorbing the momentum of the light, explains Jabez McClelland, one of the researchers. “Cooling happens if the light is tuned just below the atomic resonance and it is directed at the atoms from all directions,” he says. “Atoms moving toward the light ‘see’ photons that are Doppler-shifted closer to resonance and thus absorb more momentum in a direction opposite to their motion. The result is that they slow down. The amazing thing is that such cold temperatures (microkelvins or less) can be reached.”
The researchers explored the rather complex energy-level structure of Er, identifying five possible laser-cooling transitions (from high to low energy, they are 401, 583, 631, 841, and 1299 nm), only two (401 and 583 nm) of which had previously known rates. They also determined whether or not the transitions had “optical leaks” (a spontaneous decay to metastable states other than the ground state, which can interfere with laser cooling); the lower three transitions had virtually no optical leaks.
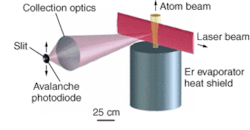
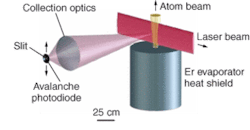
Fluorescence measurements
In an experiment, a beam of Er atoms from a heated piece of Er in a vacuum was directed into a crossed-beam laser-induced-fluorescence setup (see figure). Two transitions were studied-the 841‑nm line with an 80-mW Ti:sapphire laser, and the 631-nm line with a grating-stabilized 0.8-mW diode laser. Fluorescence measurements produced spectra identifying the characteristics of individual Er isotopes. By chopping the laser light with an acousto-optic modulator, the fluorescent lifetimes were measured; the results agreed well with calculations.
The researchers note that the large capture velocity of the 401‑nm line allows capturing of Er atoms from a thermal source. And, because the recoil velocity of the 401-nm line is smaller than the capture velocity of the 841-nm line, thermally trapped Er atoms can be further cooled by the 841-nm line. In fact, they can be cooled to 80 nK-a much lower temperature than that for other narrowband cooling atoms such as calcium, strontium, and ytterbium, making record-low laser-cooling temperatures possible.
“The colder an atom is, the longer it stays in one place, and the longer one can interact with it, the easier it is to trap-for example, with a magnetic or laser field,” says McClelland. “Erbium is particularly good for magnetic trapping because it has a very large magnetic moment. In addition, colder atoms have longer deBroglie wavelengths, making it easier to observe quantum effects. All these qualities help studies involving atomic clocks, Bose-Einstein condensation, and cold collisions, as well as any experiments where coherence matters, such as quantum information processing and quantum telecommunication.”
Laser cooling of erbium may lead to better atomic clocks, as well as single-atom doping of nanophotonic devices. The NIST researchers have already developed a single-atom doping technique for use with chromium atoms that may be applicable to Er.2
REFERENCE
1. H. Y. Ban et al., Optics Express13(8) (April 18, 2005).
2. S. B. Hill and J. J. McClelland, Appl. Physics Lett.82(18) (May 5, 2003).