Researchers at Lawrence Livermore National Laboratory (LLNL; Livermore, CA) have developed a thin, straight polymer device that takes the shape of a corkscrew when heated by a laser, and can be used to remove blood clots from stroke patients.1 The scientists believe the laser-activated device may offer a safer alternative than drugs for clot-dissolving therapy.
When a blood clot becomes lodged in the arterial network supplying the brain, the result can be an ischemic stroke that deprives the brain of oxygen and often causes permanent disability. The intravascular device developed at LLNL is designed to mechanically retrieve blood clots from arteries and restore blood flow following a stroke. In addition, it could extend the treatment window from three to eight hours after the stroke.
“This is a promising tool for treating ischemic stroke without the need for systemic infusion of thrombolytic drugs,” says Ward Small IV, one of the LLNL researchers.
The system includes a near-IR diode laser coupled to a shape-memory polymer (SMP) microactuator using an optical fiber. A thin surface layer of a platinum dye absorbs the laser light, generating heat, which causes the SMP to transform into its corkscrew shape. The SMP has the ability to resume a primary, or “memorized,” shape after being deformed into a secondary shape, explains Small. The secondary shape is maintained until the SMP is heated above its soft phase transition temperature.
Delivered through a catheter
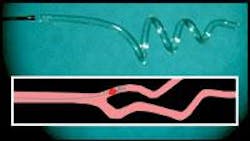
The researchers deliver the SMP microactuator in its straight rod form through a catheter near the vascular occlusion, located by fluoroscopy that is routinely used in stroke treatment. The polyurethane-based SMP device is connected to a diode laser that emits 4.89 W at 810 nm. Immersed in water at body temperature, the polymer changes shape when irradiated, from a straight rod into a 10-mm-long spiral structure with a maximum diameter of 3 mm in just three seconds . The device is then retracted and the blood clot removed from the body.
The corkscrew shape was chosen to maintain the waveguiding ability of the SMP microactuator while providing a means for capturing a blood clot (see figure). The researchers estimate that laser heating of the SMP polymer would raise the temperature of the surrounding blood by approximately 12°C, to 49°C, which can be sustained for about 15 minutes without causing permanent tissue damage.
The scientists have tested the device with an in vitro feasibility study using an artificial blood clot made of an acrylamide hydrogel inserted into an experimental water-filled model of a carotid artery with a 60° bifurcation. The polymer microactuator device was pushed into the artificial clot, activated, and used to pull back the occlusion against the flow. The scientists used thermal imaging and computer simulation to characterize the optical and photothermal behavior of the SMP microactuator.
The work was performed under the auspices of the U.S. Department of Energy by LLNL and supported by the National Institutes of Health/National Institute of Biomedical Imaging and Bioengineering. Currently, the group is discussing its technology with medical device manufacturers and plans to conduct animal studies to further evaluate the technique.
The researchers expect to make modifications including a sheath around the optical fiber for easier manipulation, radio-opacity for visualization under fluoroscopic guidance, and a method of securing the captured clot after retraction for withdrawal from the body. Based on the researchers’ previous experience with another SMP-based interventional device, they believe that human trials could begin 12 to 18 months after reaching a licensing agreement, according to Small.
REFERENCE
1. W. Small et al., Optics Express 13(20) (Oct. 3, 2005).