RAMAN SPECTROSCOPY: Raman technique improves imaging 1000 times
A new noninvasive Raman-spectroscopy technique could enable the detection of even the most minute malignant tissues if applied to human use in surgical or endoscopy applications. Research scientists in the Molecular Imaging Program at Stanford University (Stanford, CA) use a microscope that was modified to detect Raman nanoparticles to detect targets on a scale 1000 times more sensitive than is now possible using fluorescence-imaging techniques and quantum dots.1
While positron-emission tomography, optical-coherence tomography, magnetic-resonance imaging, and other bioluminescence- and fluorescence-imaging techniques are available for deep-tissue imaging, no single method simultaneously offers high sensitivity, high spatial and temporal resolution, low cost, and high throughput. And though fluorescence imaging can come the closest at limited depths, fluorescent markers often have spectral overlap, restricting the ability to interrogate multiple targets simultaneously (poor multiplexing), while background autofluorescence restricts the sensitivity and depth of the measurement and photobleaching reduces the lifetime of fluorescent markers, making long-duration analysis difficult.
Scattering allows multiplexing
Unlike fluorescence, Raman spectroscopy is a scattering phenomenon that can differentiate the spectral fingerprint of many molecules simultaneously (high multiplexing capability). Because the Raman effect is normally quite weak, surface-enhanced Raman spectroscopy (SERS)—a plasmonic effect in which molecules adsorbed onto nano-roughened noble-metal surfaces experience a dramatic increase in the incident electromagnetic field—can be used to obtain high Raman intensities comparable to fluorescence. Fortunately, particles consisting of a gold core, Raman-active molecular layer, and silica coating can be used as radiolabeled biotags to attach to particular molecules of interest within a living organism. In addition, single-walled carbon nanotubes (SWNTs) are also inherently Raman active and exhibit a strong Raman peak, allowing high-sensitivity detection. For the first time to their knowledge, the Stanford researchers have used Raman spectroscopy with both SWNT nanoparticles and silica-coated biotags (with no degradation of Raman signal detected over a five-day period) to noninvasively image small animals.
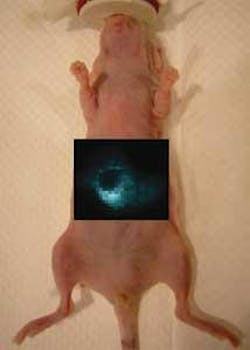
The Raman imaging setup consists of a Renishaw (Gloucestershire, England) InVia Raman microscope and a 785-nm-emitting excitation laser normally used for surface imaging. A high-numerical-aperture lens was replaced by a 12× open-field lens with a defocused beam (producing a spot size of 20 × 200 µm), to improve both illumination and light collection. A computer-controlled x-y translation stage was used to obtain raster scans for regions of interest over an anesthetized nude (hairless) mouse under test, and software algorithms were used to calculate the SERS nanoparticle concentration and generate two-dimensional mapped images of the SERS nanoparticle distribution (see figure).
In addition to imaging using SWNTs and SERS nanoparticles, multiplexed imaging for four different SERS nanoparticles (producing four different Raman spectra) was also demonstrated. “The Raman imaging strategy allows much more sensitive detection then what we have ever been able to previously achieve, primarily due to the low background signal and the enhanced Raman effect,” says Sam Gambhir, lead scientist on the study.
Because the future use of such Raman nanoparticles as SWNTs for biological studies is concerned with the toxicity and removal of these particles, researchers from the Molecular Imaging Program at Stanford also completed preliminary toxicology studies of SWNTs in a small sample of nude mice.2 The research revealed no measurable toxicity effects over a four-month period for five mice treated with SWNT particles and five mice treated with oxidized SWNTs (and both compared to a group of five untreated mice), although the researchers acknowledge that further study is needed.
REFERENCES
1. S. Keren et al., Proc. National Academy of Sciences 105(15) 5844 (April 15, 2008).
2. M.L. Schipper et al., Nature Nanotechnology 3, 216 (April 1, 2008).
About the Author

Gail Overton
Senior Editor (2004-2020)
Gail has more than 30 years of engineering, marketing, product management, and editorial experience in the photonics and optical communications industry. Before joining the staff at Laser Focus World in 2004, she held many product management and product marketing roles in the fiber-optics industry, most notably at Hughes (El Segundo, CA), GTE Labs (Waltham, MA), Corning (Corning, NY), Photon Kinetics (Beaverton, OR), and Newport Corporation (Irvine, CA). During her marketing career, Gail published articles in WDM Solutions and Sensors magazine and traveled internationally to conduct product and sales training. Gail received her BS degree in physics, with an emphasis in optics, from San Diego State University in San Diego, CA in May 1986.