Optically pumped xenon yields highly sensitive NMR biosensor
Scientists once thought of the nuclear-magnetic-resonance (NMR) chemical shift as a mild annoyance in their attempts to measure magnetogyric ratios for nuclei. Now they are using techniques, such as optical pumping, to enhance detection sensitivity for a broad range of sensing applications.
Researchers in the Alexander Pines and David Wemmer groups at Lawrence Berkeley National Laboratory (LBNL; Berkeley, CA) and the Peter Schultz group at Scripps Research Institute (La Jolla, CA) have collaborated to create a new biosensor molecule based on optically pumped xenon (Xe) that has the potential to provide a much more flexible approach to complex bioassays than can be obtained through existing direct optical methods relying on fluorescence.1 While fluorescence-based probes are highly sensitive, the variety of measurements that can be taken simultaneously is limited by spectral overlap. Also, the need for direct laser stimulation severely limits their use for in vivo observations.
In contrast, NMR spectroscopy offers a biologically noninvasive approach with a potentially broad capacity for parallel measurements, because slight differences in the chemical environment of atomic nuclei within a static magnetic field show up as distinct chemical-shift values. Increasing the sensitivity of NMR in detecting these differences is key to making the method practical in the area of biosensing, because the spin-population difference in nuclei is only 1 in every 100,000 detectable spins at generally available magnetic-field strengths.
Introducing optically pumped noble gases can boost this NMR sensitivity by three to four orders of magnitude, said Janette Ruiz, a graduate student in the LBNL research laboratory of Alexander Pines. The Berkeley-Scripps research team has adapted optically pumped Xe—with a chemical shift range in excess of 7500 parts per million—to protein/ligand biosensing applications and have obtained spectroscopic sensitivities on the order of tens of nanomoles. In addition to its large laser-induced chemical-shift sensitivity, Xe is particularly well-suited for biosensing applications because it is a nontoxic element that retains its laser-induced polarization for relatively long time periods on the order of 40 min.
Optical pumping
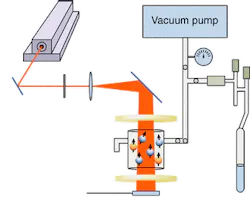
The actual laser-polarization process must be performed in two steps, Ruiz said. First, angular momentum from the circularly polarized beam of a continuous-wave, 795-nm-emitting laser diode polarizes the spin populations of valence electrons in rubidium (Rb) gas atoms. Then, the polarization is transferred through a spin-exchange process to nuclei in Xe gas. The spin-exchange process is based on a hyperfine interaction in which magnetic fields generated by moving electrons interact with magnetic-dipole moments of adjacent atomic nuclei, thereby influencing nuclear spin polarizations.
The gaseous mixture in this process only contains about 1% Xe, however, and only a small amount of Rb vapor emanating from less than half a gram of liquid Rb heated to about 150°F, Ruiz said. An 89% helium concentration raises the pressure of the mixture to 5 atmospheres and increases optical sensitivity of the Rb by more than an order of magnitude through broadening the wavelength response by a few tenths of a nanometer on either side of the 795-nm peak. A 9% nitrogen concentration maintains the spin polarization of Rb electrons by collisionally quenching electrons that spontaneously lose their polarization prior to spin exchange with the Xe nuclei (see Fig. 1).
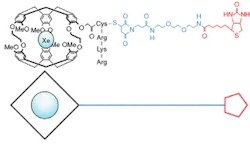
Converting the optically polarized Xe into a functional biosensor involved synthesizing a molecule to bind the Xe to various proteins of interest. The molecular model that was developed consisted of three parts: a hollow cryptophane molecule, effectively acting as a cage for the Xe; a biomolecule-targeting ligand, in this case the commercially available biotin or B-vitamin ligand; and a polymer-linking assembly to connect the cage with the ligand while amino acids between cage and linking assembly make the entire molecule water-soluble (see Fig. 2).The researchers validated the concept experimentally by targeting the avidin protein from egg white and obtaining sensitive chemical shift information related to the resulting binding event between protein and ligand. They also noted variations in chemical shift based on factors such as the molecular structure of the cryptophane cage as well as chirality of the linking chain and ligand. Manipulating these types of variables has the potential to provide the desired breadth of simultaneous measurements that are not easily achieved with fluorescent methods. Patents are pending on the novel NMR-based Xe biosensing technology, which has also been made available for licensing through the Technology Transfer Department at LBNL.
REFERENCE
- M.M. Spence et al., Proc. Natl. Acad. Sci. USA 98(19) 10654 (Sept. 11, 2001).
About the Author
Hassaun A. Jones-Bey
Senior Editor and Freelance Writer
Hassaun A. Jones-Bey was a senior editor and then freelance writer for Laser Focus World.