Mesh paves the way for higher-sensitivity spectroscopy
You can only get so much light through a hole—unless it is a very small hole and you can access surface plasmons. James Coe at Ohio State University (Columbus, OH) and coworkers have found metal meshes that transmit more IR light than is incident upon the holes.1, 2 The phenomenon reveals a thousandfold enhancement in the light absorbed by one-molecule-thick coatings. The increased light has a number of potential applications, including higher-sensitivity surface spectroscopy.
The researchers were investigating meshes as a scaffolding for nanotechnology. In 1988, researchers at NEC Research Institute (Princeton, NJ) noticed that nanometer-sized arrays of silver could transmit an extraordinary amount of visible light in the form of energy packets called surface plasmons.3 A surface plasmon consists of collectively excited electrons at the surface of a metal.
Instead of silver, Coe's group used commercially available nickel mesh, and instead of visible radiation, they used IR light to match with molecular vibrations. For surface plasmons to be formed, the spacing between the holes in the mesh must be comparable to the wavelength of the light hitting the mesh. The surface-plasmon lifetimes can be increased, however, if the holes are coated with copper. So they developed methods of coating the mesh that filled in the edges of the holes. They soon discovered that they could tune the process to shrink the holes to whatever size they wanted.
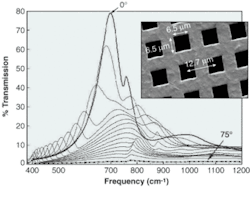
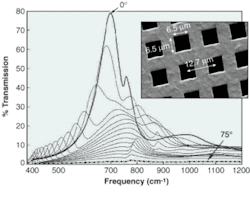
Because the holes normally cover only 25% of the surface, one might expect the mesh to transmit, at most, 25% of incoming light. But in tests, the coated mesh transmitted 75% of the light at the peak wavelength, which suggested that light falling on the metal is guided along the surface and coupled into the microchannels by surface plasmons (see figure).
The researchers removed most of the non-plasmon-related light by decreasing the area through which light incident on the holes could pass straight through the mesh. They doubled the mesh, thus reducing the open area to 10%—and saw 60% transmission. When they stacked four meshes (providing about 0.5% open area), the transmission was 22%.
A layer of fat
Like photonic-bandgap crystals and many other nanotechnology advances, the key to accessing the plasmons appears to be controlling the size of the holes. Coe's group investigated coating the mesh with copper, and with a self-assembling monolayer of 1-dodecanethiolate (a long-chain fat) and phospholipids (cell membrane material with long-chain fats).
"With the right coating process, we found we could precisely control the diameter of the holes, and the amount of light transmitted. In effect, the coated mesh acts like an optical switch," Coe said. "With the addition of heat controls, you could call it a thermo-optical switch." The university has filed two patent applications for the technology and is looking for commercial partners to develop it further.
The phase of the fat molecules can be used to control the amount of light passing through because of their shape, Coe said. "At room temperature, the molecules form long chains that stand straight up on the surface of the mesh. As the temperature rises, the chains melt, changing the polarization of light throughout the hole," he said. The effect can be repeated as long as the molecules are kept hydrated.
When this arrangement of materials (a mesh full of subwavelength channels) is used for spectroscopy, the dramatically longer path lengths result in higher sensitivity. Instead of the path length being about twice the thickness of the coating (as it is in reflection-absorption experiments), it becomes the length of the microchannel plus whatever distances the plasmons run along the front or back surfaces of the mesh. (Stacking the meshes also adds to the path length.) In practice, the researchers say, this could amount to a thousandfold enhancement in the fraction of light absorbed, allowing very sensitive detection schemes using vibrational spectroscopy to detect molecular surface species.
REFERENCES
- J. V. Coe et al., posters 365 and 366, American Chemical Society national meeting (Sep. 10, 2003, New York).
- S. M. Williams et al., J. Phys. Chem. B, accepted for publication.
- T. W. Ebbesen et al., Nature 391, 667 (1998).