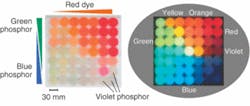
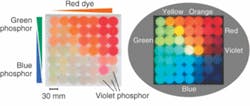
Illumination consumes no electricity
While bright, long-duration phosphorescence has been achieved in the violet-to-green wavelength range with rare-earth-doped strontium aluminum oxides, attempts to achieve orange-to-red phosphorescence using various metal-doped oxides have produced results inferior in brightness and afterglow duration. But researchers at Ryukoku University (Otsu, Japan) have demonstrated conversion of weak phosphorescence to efficient strong fluorescence of a different color using a different approach-downconversion of organic dyes-and have now fabricated acrylic resins that yield bright illumination across the entire visible spectral range without consuming electricity.
To fabricate the resins, phosphor particles and dye molecules were suspended in a photocurable acrylate matrix and solidified. Depending upon the phosphor and dye molecule concentrations, emission of color across the visible spectrum could be obtained after exposure to a white-light source. Upon removal of the light source, illumination decayed from approximately 1 nW/mm2 to around 0.01 nW/mm2 after one hour. Optical power of the red emission-obtained by mixing blue and green phosphors with red organic dye-measured 90 minutes after the excitation source was turned off was found to be seven times higher than that of conventional red phosphors. Contact Mitsunori Saito at [email protected].