LASER ISOTOPE SEPARATION: Silex uranium-enrichment scheme may not be mature enough for practical use
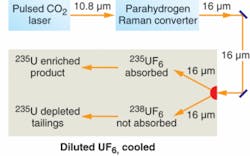
The laser isotope-separation process called Silex may look good to General Electric (Wilmington, NC) for enriching uranium-235 (U-235) concentration to the levels required in nuclear reactors (see www.laserfocusworld.com/articles/266374), but it does not appear mature enough to enrich U-235 concentration to the higher levels needed for nuclear weapons, according to a team that reviewed the Silex process for the International Atomic Energy Agency (IAEA).
Australian scientists Michael Goldsworth and Horst Struve developed the process, and from 1996 to 2002 received support from the United States Enrichment Corp. (Bethesda, MD); the two scientists have since formed a public corporation, Silex Systems (Lucas Heights, NSW, Australia). Last year they licensed the Silex process to General Electric.
The process is based on selective excitation of uranium hexafluoride (UF6) molecules that contain U-235 by laser light at a narrow spectral line near 16 µm, but few details have been released (see figure). The Los Alamos National Laboratory (Los Alamos, NM) initially explored the concept three decades ago, but the U.S. Department of Energy later abandoned it in favor of atomic-vapor laser isotope enrichment.
A November 2004 report from Greenpeace warned that the Silex process could encourage nuclear proliferation by simplifying the difficult process of uranium enrichment.1 But a 2005 report from Los Alamos, recently made available by the Federation of American Scientists, raises doubts about that claim.2
Silex “is not currently capable of producing significant amounts of HEU (highly enriched uranium),” wrote John L. Lyman, who visited Silex Systems with three IAEA representatives in February 2005, when he was working at Los Alamos. He cited a number of important processing limitations.
Limited wavelength- conversion output
One problem is the limited optical output available on the 16 µm line, generated by Raman shifting 10.8 µm output from a pulsed carbon dioxide (CO2) laser in a high-pressure parahydrogen cell. The CO2 lasers can generate 1 J pulses, but only at a limited repetition rate, and only a fraction of the pulse is in the pump band. Unspecified “additional nonlinear optical tricks” are needed to convert the CO2 pump light to the correct wavelength to pump the Raman cell. The lasers are 1% efficient and the Raman conversion 25% efficient, so the overall efficiency is 0.25%.
Operation of the CO2 lasers at their current 50 Hz repetition rate is not sufficient to produce significant enrichment, according to Lyman. “A mature facility would have the capability to increase that rate by more than an order of magnitude,” he wrote. The problem is not just that higher repetition rates are needed to increase the amount of uranium enriched. “At low rates, the process does not work because of large amounts of unprocessed material in the product stream,” Lyman wrote. “At 50 Hz, only 1% of the material in the feed stream is processed, with 99% going unprocessed, limiting the possible amount of enrichment.”
Another issue is the limited amount of material that can be processed at once. The vapor must be cooled to ensure good separation of the absorption lines of the two isotopes. But under those conditions, Lyman wrote, “rapid catastrophic condensation occurs if the UF6 molecular density gets too high.” In his experience, that maximum density is 1015 molecules per cubic centimeter.
With many details classified or proprietary, it is hard to quantify the processing. Lyman wrote that if a laser could illuminate a one-liter volume at an ideal repetition rate, it would take about 100 hours to produce one kilogram of U-235-assuming complete separation of the U-235 and U-238 isotopes. However, most processes require multiple stages of separation, and according to Lyman’s comments, a 5000 Hz laser would be needed to process all the feed stream (a mixture of UF6 and an unidentified diluting gas).
REFERENCES
1. Secrets, Lies, and Uranium Enrichment, the Classified Silex Project at Lucas Hill, Greenpeace, November 2004
2. J.L. Lyman, Enrichment separative capacity for SILEX, Los Alamos National Laboratory (LA-UR-05-3786); posted at www.fas.org/sgp/othergov/doe/lanl/docs4/silex.pdf.
About the Author
Jeff Hecht
Contributing Editor
Jeff Hecht is a regular contributing editor to Laser Focus World and has been covering the laser industry for 35 years. A prolific book author, Jeff's published works include “Understanding Fiber Optics,” “Understanding Lasers,” “The Laser Guidebook,” and “Beam Weapons: The Next Arms Race.” He also has written books on the histories of lasers and fiber optics, including “City of Light: The Story of Fiber Optics,” and “Beam: The Race to Make the Laser.” Find out more at jeffhecht.com.
