TWO-PHOTON FLUORESCENCE: Fluorescent dyes recover after eight hours of rest
A broad range of applications that include optical switches for telecommunications, high-power light sources, and numerous optical-fiber and display devices could benefit from more-robust optical materials that either do not degrade or degrade reversibly upon prolonged exposure to light. Researchers at Washington State University (WSU) have encountered two dye molecules—Disperse Orange 11 (DO11) and a material made by the U.S. Air Force (AF455)—that recover spontaneously from photodegradation if left in the dark for a matter of hours.
Previous work had demonstrated recovery of photodegraded dyes in a solution in which mobility of fresh molecules enables replacement of depleted ones. The WSU work, however, was done in a dye-doped polymer. “Measuring materials in a liquid is simpler,” said Mark Kuzyk, who leads the WSU research effort. “But for devices you need material in a medium that does not flow.”
Unexpected effect
Nonlinear optics of organic materials and fiber optics in polymer fibers are two areas of focus in Kuzyk’s laboratory for nonlinear optical studies. The researchers were initially looking for materials with strong nonlinear optical characteristics. While comparing two-photon absorption properties among different materials, they noticed that a photodegraded sample of the chromophore AF455 doped into poly[methyl methacrylate] (PMMA) fiber recovered its two-photon fluorescence properties after being left in the dark overnight.
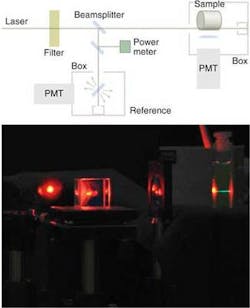
The researchers previously noticed a similar recovery of amplified spontaneous emission efficiency in a photodegraded sample of DO11 but had dismissed the observation as an anomaly of that particular dye. A similar effect in AF455, later, led them to reconsider and investigate the phenomenon directly. They did so by exposing the AF455 sample to a laser source to stimulate two-photon fluorescence, which they recorded using a photomultiplier tube (PMT) connected to a digital oscilloscope.
Photodegradation of the sample occurred at a rate that turned out to be proportional to the intensity of the source. After three hours, laser illumination was discontinued and the sample was left in the dark for eight hours. During this time the sample was momentarily illuminated once every 30 minutes for a period of 30 seconds to monitor recovery of two-photon fluorescence. During the eight-hour period the sample was exposed to the laser for a total of only four minutes.
The light source was a tunable optical-parametric-generator laser pumped with a frequency-tripled Nd:YAG laser to produce 25 ps pulses, at 800 nm in one experiment and at 828 nm in another, with a 10 Hz repetition rate. The wavelengths were chosen to fall between 790 and 830 nm, a range in which AF455 exhibits relatively high two-photon fluorescence (see figure).
During the constant illumination portion of the experiment, with the most intense dose (4.5 µJ per pulse), within 200 minutes the fluorescent response of the sample dropped to about 60% of the starting level. The sample began to recover just half an hour after being placed in the dark and continued to recover throughout the eight-hour dark period. The more intense the laser used, the faster the material lost its ability to respond. Regardless of the intensity of light used to exhaust the material, however, recovery proceeded with the same time constant, indicating that the mode of recovery does not depend on the severity or means of the decline, Kuzyk said.
The self-healing property does not appear to be a general phenomenon of fluorescing materials. Kuzyk’s team is working to understand the mechanism in hopes of finding ways to endow other molecules with the same ability. In DO11, the photodegradation seems to involve molecules pairing up to form dimers. Recovery seems to occur when the molecular pairs disassociate. The larger AF455 molecule does not seem to photodegrade and recover through formation and disassociation of molecular pairs, however, and the researchers have yet to propose a model for it.
REFERENCE
1. Y. Zhu, J. Zhou, M.G. Kuzyk,Optics Lett. 32(8) 958 (April 15, 2007).
About the Author
Hassaun A. Jones-Bey
Senior Editor and Freelance Writer
Hassaun A. Jones-Bey was a senior editor and then freelance writer for Laser Focus World.