PHOTONICS APPLIED: GREEN PHOTONICS: Harnessing sunlight moves beyond conventional photovoltaics
Crystalline silicon solar cells have been converting sunlight to electricity for decades, with conversion efficiencies exceeding 20%. But decades of materials and photonics advances happening in parallel are enabling researchers to push the conversion of sunlight to energy beyond conventional photovoltaics. Not only are they using advanced thin-film materials, glass doping methods, and concentrating optics to improve sunlight collection efficiencies beyond the limits of silicon, but they are also developing new ways to harness the power of the sun: A research consortium converts sunlight into its fundamental spectral components toward a 50% efficient solar cell; building-integrated photovoltaics (BIPV) convert plate-glass windows into transparent solar panels that can double as light fixtures; and researchers in artificial photosynthesis tap the power of the sun for hydrogen fuel generation.
Not your father's photovoltaics
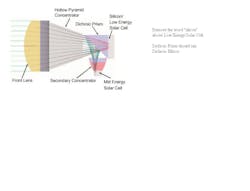
When people think of photovoltaic (PV) technology, they typically think of conventional flat-plate crystalline silicon solar panels. Although solar conversion efficiencies have steadily risen over the past few decades to levels now exceeding 20%, flat-plate silicon PV has new rivals. In addition to various concentrating photovoltaic (CPV) architectures, a result from the Very High Efficiency Solar Cell (VHESC; www.darpa.mil/sto/smallunitops/vhesc.html) program described conversion efficiencies just above 36% at EUPVSEC in Hamburg late last year for its PV technology based on spectrally sorting concentrated sunlight.1 The report on the new spectral splitting approach (from a very large group of collaborators) was led by the University of Delaware (Newark, DE), with critical testing done by the National Renewable Energy Laboratory (NREL; Golden, CO) and a co-author from Arizona State University (Tempe, AZ). The VHESC solar cells capture sunlight using a double-convex concentrating lens with magnesium-fluoride (MgF2) coating and a 30 mm2 aperture followed by a dichroic beamsplitter that reflects light at wavelengths shorter than 870 nm and sends them to a gallium-indium-phosphide/gallium-arsenide (GaInP/GaAs) detector, while transmitting light at wavelengths longer than 870 nm and sending those to a GaInAsP/GaInAs detector (see Fig. 1). The concentration is approximately 30X when using 1 mm2 detectors. Already, a sum of the solar cells' efficiency has reached 42.7% for the VHESC wavelength-optimized detection scheme.
But despite their potential for ultrahigh efficiencies and long lifetimes, flat-plate and even CPV options are limited to ground and roof-mounted configurations. Fortunately, decades of materials research has enabled a host of thin-film PV technologies that are lightweight and potentially less expensive, and offer new consumer options that flat-plate PV cannot address. Not only do thin-film PV products made from exotic semiconductor materials such as cadmium telluride (CdTe) and copper indium gallium diselenide (CIGS) have conversion efficiencies approaching 20%, their method of manufacture lends itself to roll-to-roll production or printing-based processes, facilitating flexible, moldable, or spray-on solar-cell products. For example, a cylindrical PV option from Solyndra (Fremont, CA) consists of thin-film CIGS materials deposited on an inner glass tube encapsulated within an outer glass tube to prevent moisture ingress. The cylindrical shape gathers sunlight from all angles throughout the day, increasing photon collection without the need for angle mounts or expensive tracking systems.
Also in the thin-film category are even less expensive organic or polymer-based PV materials with efficiencies now approaching 10%. No longer do PV materials simply sit on the ground or roof; they are lightweight and can be incorporated into textiles or molded into architectural materials like roof tiles—an example of BIPV.
Konarka's (Lowell, MA) Power Plastic, an organic photovoltaic (OPV) product, converts indoor or outdoor light into direct-current electricity and is sold to OEMs for incorporation into solar-gathering products such as a 70 × 34 cm thin, flexible, and portable power charger (for batteries or small electronics) that operates between -20º and +65ºC and provides 3.9 W or 7.9 V at 1 Sun (full outdoor sunlight) with a specified lifetime of 3–5 years. It consists of a photoreactive printed polymer layer, a transparent electrode layer, a plastic substrate, and a protective packaging layer (all recyclable materials) with total thickness of 0.5 mm and offered in up to 60 in. widths at almost any length, thanks to roll-to-roll processing. A 2008 NanoMarkets (Glen Allen, VA) report says that plastic solar panels are expected to generate revenues of $80 million by 2015 in the consumer and portable electronics market alone.
Imagine sitting under a Power Plastic-covered umbrella sipping a drink while charging your cell phone and digital camera, camping out in your Power Plastic-covered tent to keep your battery-powered radio or lantern in working order, or mounting Power Plastic on your carport roof to trickle-charge your electric car. Not all of these innovations are available yet, but you can see Konarka's Power Plastic in action as a BIPV material on the wavy roofs of San Francisco's Municipal Transportation Agency (MTA) transit shelters (see Fig. 2). By 2014, 300 of these shelter covers will be installed in sunny locations, powering LED lighting, "next bus" LED signage, and Wi-Fi routers, and feeding an estimated 43,000 kilowatt hours (kWh) of electricity annually back into that city's grid over an estimated 25 year lifespan.
While some BIPV products are in development (such as Konarka's "curtain walls" that act as structural elements for a building façade), wearable PV textiles with the somewhat soft consistency of clothing are still in the research stage.
Windows to the future
Can you imagine BIPV materials so thin and transparent that they can be applied to ordinary windows and harness energy from the sun? You're not alone: Companies like Solarmer Energy (El Monte, CA), and Rainbow Solar (RSi; Pasadena, CA) are already commercializing solar-collecting thin films for ordinary windows.
In late 2009, Solarmer achieved a record-breaking NREL-certified 7.9% conversion efficiency for its polymer OPV solar cell. And just recently, Solarmer and Joel Berman Glass Studios (Vancouver, BC, Canada) jointly developed a series of BIPV decorative glass panels that generate electricity (see Fig. 3). The semitransparent PV panels consist of textured kiln-cast glass laminated to Solarmer's transparent plastic solar cells. Each panel will convert sunlight to energy and look good doing it.2
"OPV is the first solar technology that has the capability to generate electricity at a cost on par with conventional fuels, making it a cost-effective alternative energy source even without government subsidies," says D'Juvayne Christian, marketing communications coordinator for Solarmer Energy. "Solarmer's panels could potentially drive the cost of electricity generated by the sun down to 12–15 cents/kWh with direct manufacturing costs less than $1/W. For widespread market acceptance of this technology, product lifetimes of three to five years and efficiencies in the 8–10% range are needed."
One limitation of PV polymers is their limited absorption in the solar spectrum and 100–200 nm thinness, which results in a significant amount of unabsorbed light. Even at the peak absorption wavelength, transmittance is only 60–70%. However, Solarmer transforms this limitation into a benefit by sandwiching its very thin polymer active layer between two proprietary transparent electrodes. In this cell, the electrodes are truly transparent throughout the film surface, enabling transmittance as high as 85% and opening up new markets such as power windows. Using their portfolio of technology developed in a joint effort between the University of California–Los Angeles (UCLA) and the University of Chicago, Solarmer has moved from lab prototypes to full roll-to-roll pilot-scale manufacturing. The company plans to deliver production-grade samples to potential customers by the end of 2010, and to start selling its panels in early 2011.
Another contender in the PV window market is Rainbow Solar or RSi, which announced the first production BIPV semitransparent glass window that generates up to 250 W of electrical power from windows as large as 9 ft2. As well as blocking 50% of sunlight to reduce heating and cooling needs, the multilayer window can be designed to do double duty and emit light, acting as an evening light fixture.
Learning from nature
In addition to photosynthesis-inspired dye-based solar cells and a new dendrimer-based solar cell that channels photons from a nonbiological organic molecule to a polymer wire attached to an electrode for photon-to-electron conversion, perhaps the ultimate use of biomimetics is artificial photosynthesis. Just as plants use sunlight and water to convert carbon dioxide to sugars and oxygen, artificial photosynthesis uses a photoelectrochemical cell to split water into hydrogen and oxygen (the opposite of a fuel cell in which hydrogen and oxygen combine to produce water and energy) that can be stored for future use.3
In the photoelectrochemical cell, a photoactive semiconductor forms a junction in contact with a liquid or solid electrolyte. Upon illumination, electron-hole pairs are produced in the photoactive material and drive a chemical reduction and oxidation, leading to hydrogen and oxygen production, respectively. Here, the photon energy is converted directly into chemical energy in the form of hydrogen. "Artificial photosynthesis and related research is the most important area for making the best of solar power, as it enables us to store the energy efficiently, ultimately in the form of hydrogen fuel that can also be used for mobile vehicles," says Mae-Wan Ho, director of the Institute of Science in Society (ISIS; London, England; www.i-sis.org.uk). "It is not yet close to commercialization; but with proper investment, it could be, say, in ten years' time."
Just recently, researchers from the Massachusetts Institute of Technology (MIT; Cambridge, MA) have mimicked how natural pigments in plant cells absorb sunlight.4 The researchers engineered a harmless bacterial virus called M13 that attracts and binds with molecules from a catalyst and a biological pigment to promote the water-splitting reaction that creates hydrogen and oxygen. While the method is effective in four-fold production of oxygen, a means is still needed to combine the protons and electrons produced back into hydrogen; in addition, a less expensive catalyst other than iridium is being sought.
An alternate technique from researchers at The Weizmann Institute of Science (Rehovot, Israel) replaces iridium with an organic/metal ruthenium complex that is used in a series of heat- and light-activated reactions to liberate hydrogen and oxygen from water, without using up the catalyst; apparently, the light-activated step produces oxygen while also causing the ruthenium complex to revert to its orginal state.5 "This is a completely new approach toward light-driven water splitting," says David Milstein, Weizmann Institute Israel Matz professorial chair of organic chemistry. "However, it is at a fundamental stage, and requires additional scientific advances before it can become technologically feasible." Such a caveat is placed on nearly all sunlight-conversion research; after all, solar cells are just now gaining momentum some 50 years after their invention. Here's hoping that it doesn't take another 50 years (and a few more energy crises) before these next-generation technologies enter the marketplace.
REFERENCES
- A. Barnett et al., Progress in Photovoltaics: Research and Applications 17, 75–83 (October 2009).
- D. Christian, "Solarmer, Inc.," OSADirect Magazine 2, 15–18 (September 2009).
- M.-W. Ho, "Harvesting Energy from Sunlight with Artificial Photosynthesis," Science in Society 43 (Autumn 2009).
- Y.S. Nam et al., "Biologically templated photocatalytic nanostructures for sustained light-driven water oxidation," Nature Nanotechnology online Letter (Apr. 11, 2010).
- S.W. Kohl et al., Science 324, 5923, 74–77 (Apr. 3, 2009).
About the Author

Gail Overton
Senior Editor (2004-2020)
Gail has more than 30 years of engineering, marketing, product management, and editorial experience in the photonics and optical communications industry. Before joining the staff at Laser Focus World in 2004, she held many product management and product marketing roles in the fiber-optics industry, most notably at Hughes (El Segundo, CA), GTE Labs (Waltham, MA), Corning (Corning, NY), Photon Kinetics (Beaverton, OR), and Newport Corporation (Irvine, CA). During her marketing career, Gail published articles in WDM Solutions and Sensors magazine and traveled internationally to conduct product and sales training. Gail received her BS degree in physics, with an emphasis in optics, from San Diego State University in San Diego, CA in May 1986.