Direct-write laser processing creates tiny electrochemical systems
Advances in integration and processing techniques have significantly reduced the size of microelectronic and microelectromechanical devices. Laser direct-write can fabricate micro power systems compatible with these microdevices.
In recent years, advances in integration and processing techniques have enabled microelectronic and microelectromechanical devices to shrink in size dramatically. While these new devices have been used as components in larger sensor, actuator, and control systems, the lack of commensurately sized power sources has limited their ability to function autonomously. One solution to this challenge is to integrate micro power sources directly into the microdevice package, saving valuable weight and space. Ambient-condition, laser direct-write processes make this vision a reality.
Common electrochemical systems for power generation, such as ultracapacitors, batteries, or solar cells, comprise three main internal components: the negative electrode or anode, positive electrode or cathode, and electrolyte/separator. Each of these is typically constructed of materials with a large degree of structural complexity, such as nanocomposites, solid-state polymers, liquids, or mesoporous mixtures of electrochemically active materials. The materials for micro power sources are no different, but the unique challenge is to maintain their electrochemical and structural integrity in a confined space subject to the limitations, such as temperature and pressure, imposed by the microdevices, their substrates, and packaging.
Furthermore, to meet the power demands of a given microdevice, combinations of different types of cells (hybrid power systems) are needed.1 For instance, an application may require constant low power (lithium or alkaline microbattery) or an occasional short burst of energy (ultracapacitor or capacitor), as well as the ability to be recharged (solar cell). Laser direct-write processes provide a general set of processing tools that can rapidly prototype and fabricate these different electrochemical energy storage components. These processes can even embed the electrochemical components directly within the substrates, reducing the size of the entire microdevice package even further.
Laser direct-write
Laser direct-write is a general term that encompasses modification, subtraction, and addition processes capable of creating patterns of materials directly on substrates without the need for lithography or masks. Given a laser source, one can either fix the location of the beam and raster the substrate using motion-control stages, fix the substrate and raster the beam using mirrors, or a combination of both. For laser direct-write modification (LDWM) or subtraction (LDW-), the material of interest is directly irradiated and is either removed (laser micromachining) or modified (melting, sintering, and so on). In both cases, either pulsed or CW lasers can be effective.
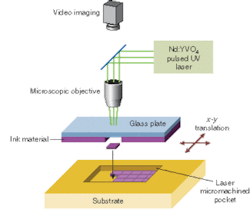
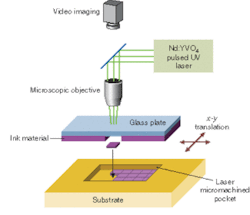
Our method of laser direct-write addition (LDW+) begins with the material of interest in powder form and combines it with a liquid carrier to form an ink (see Fig. 1). This ink is spread on a glass plate fixed approximately 100 µm from the substrate. A pulsed UV laser irradiates the ink from behind the glass plate to propel a mass of material forward onto the substrate below. This laser printing process takes place by rastering either the beam or the substrate to produce a pattern of material. Originally LDW+ was developed to deposit "electronic-grade" materials such as metals for interconnects, cermets for resistors, and dielectrics for capacitors, but it quickly became evident that the porous and wet nature of the as-deposited materials were not optimal for these types of systems.2
Fortunately, the same features of the deposited materials that make laser direct-write less than perfect for electronic grade materials, make it ideally suited for creating mesoscale patterns of the electrochemically active materials for micro power sources. The wet, porous structures that are present after LDW+ exhibit extremely large surface areas that allow more complete contact between the electrodes and the electrolyte and therefore enable better charge transfer and a more complete utilization of the electrode materials. These ambient processes can attain LDW+ resolutions down to about 25 µm, while LDW- and LDWM resolutions can be as small as 1 to 5 µm. In contrast to most other direct-write deposition techniques available, LDW+ is able to print highly acidic, caustic, or other chemically reactive materials, an important consideration for these electrochemical systems.
These laser direct-write processes should not be confused with typical thin-film material processing such as vacuum or chemical vapor deposition. Films deposited by LDW+ can be made arbitrarily thick by simply adding layers of material in the same location (typical thin-film processes become difficult above 1 to 5 µm). This approach can place a larger mass of active material in a fixed footprint, yielding higher energy and power densities than can be made with conventional approaches. Furthermore, the laser direct-write technique is not chemistry specific so that the different materials required for ultracapacitors, microbatteries, or solar cells can all be accommodated in the same "tool" by simply changing the ink.
The most important aspect of laser direct-write processes in our application is that they do not harm the electrochemical or structural properties of the transferred materials. Microstructural characterization by SEM and x-ray diffraction shows no difference between the starting powder and the laser-transferred material and neither does electrochemical characterization by cyclic voltammetry or chronopotentiometry. We attribute the successful deposition of materials to the proposed underlying mechanism that the laser energy is absorbed in a small region near the ink/glass interface, generating a local vaporization of material. Expansion of this vapor causes the majority of the ink to be propelled forward although it has not directly encountered the incident laser. This same "gentle" mechanism successfully allows living cells to be deposited unharmed to a substrate below.
Planar structures
Conceptually, the simplest system to construct is one in which the electrodes of the electrochemical cell are arranged adjacent to each other. For instance, an ultracapacitor or supercapacitor is a high-power-density component with a large specific capacitance whose voltage depends linearly on the stored charge, as in a standard capacitor. The chemistry of one such system (hydrous ruthenium oxide) is such that its two electrodes are identical to each other. Thus, we use LDW+ to deposit a uniform layer of the hydrous ruthenium oxide powder mixed with sulfuric acid electrolyte. LDWM and LDW– process the deposited material to isolate two electrode pads that are separated by a small (20-µm) gap (see Fig. 2). Overall size and mass of these systems is exceedingly small at less than 100 µg, which is less than the mass of an eyelash. Recent progress with LDWM has enabled us to devise methods of separating the electrodes and refilling the gap with liquid electrolyte in a single processing step.
Planar alkaline microbatteries represent the next level in complexity since they require two different materials for the positive and negative electrodes. We have developed a standard alkaline chemistry of zinc anode, silver oxide cathode, and potassium hydroxide (KOH) electrolyte. By mixing the electrode materials with KOH electrolyte we use LDW+ and LDW- to create various planar geometries such as noncompact, interdigitated, or circular structures. A droplet of KOH is added to the system after fabrication to fully activate the cell.
Stacked structures
Although planar structures are relatively easy to construct, stacking the electrodes on top of each other provides more efficient use of limited material and space. These structures require materials that are rigid enough to support the upper layers but their electrochemical properties must not be compromised. To this end, we have focused on the rechargeable lithium-ion microbattery system, which presents many unique challenges to developing an ambient process that will not harm the sensitive chemistry.
The key ingredient in these systems is a novel hydrophobic nanocomposite solid polymer ionic liquid (c-SPIL) separator material that has high-ionic conductivity and is chemically and structurally stable. We used LDW+ to deposit sequential layers of lithium cobalt oxide or lithium manganese oxide (LiCoO2 or LiMnO4) cathode, c-SPIL, and carbon anode into a laser micromachined pocket on a thin polyimide substrate (see Fig. 3).
The missing link for the development of a fully integrated micro power source is the ability to harvest energy from the environment to replenish the limited power supply of the microbatteries and ultracapacitors. Using the same combination of laser-based processes, we have started developing nanoparticle titanium dioxide (TiO2)-based solar cells. LDWM allows local sintering of nanoparticles at low-substrate temperatures to enable a large carrier lifetime without destroying the high-surface-area mesoporous structure.
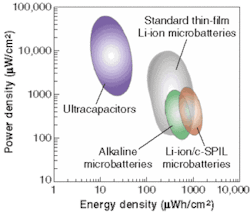
The detailed electrochemical results of the different systems fabricated by laser direct-write processing can be found in the literature for ultracapacitors, alkaline microbatteries, lithium-ion microbatteries, and solar cells.4 Our results for the different systems show that the energy density from both microbattery chemistries is comparable with the maximum values reported in the literature for lithographic and vacuum fabrication techniques (see Fig. 4).5, 6 Current work focuses on increasing both power and energy density through optimization of the materials and laser processes.
Beyond integration
There is a demand for small-scale power systems that are compatible with current and future advances in microdevice technology. The use of laser direct-write processes to fabricate micro power systems can satisfy this demand. These ambient and low-temperature techniques provide fully embedded solutions, which enable integrated power sources within most types of rigid and flexible substrates. The ability to process a wide range of structurally and chemically sensitive materials in complex 2-D and 3-D patterns is not limited to micro power sources; it could also significantly benefit applications in catalytic, sensor, or biomaterials. The extension of laser processing to nontraditional areas such as these opens doors to new possibilities for advanced devices in a wide variety of fields and will have a lasting impact on future technology.
ACKNOWLEDGMENTS
This work was supported in part by the Office of Naval Research and the National Research Council. The authors would like to thank the contributions made by Mike Ollinger and Ray C.Y. Auyeung to this research effort.
REFERENCES
- Koeneman, P.B. et al, J. Microelectromech. Sys. 6, 355 (1997).
- A. Piqué and D. B. Chrisey, Direct Write Technologies for Rapid Prototyping Application, Academic Press (2001).
- D. B. Chrisey et al., Chem. Rev. 103, 553 (2003)
- C. B. Arnold et al., J. Electrochem. Soc. 150, 571 (2003); C. B. Arnold et al., Appl. Phys. A., In Press (2004); T .E. Sutto et al., Submitted to Nature (2004); H. Kim et al., Submitted to Appl. Phys. Lett. (2004).
- N. J. Dudney and Y. I. Jang, J. Power Source 300, 119, (2003).
- P. H. Humble et al., J. Electrochem. Soc. 148, 1357 (2001).
CRAIG B. ARNOLD is an assistant professor in mechanical and aerospace engineering at Princeton University, (Princeton, NJ); THOMAS E. SUTTO is a research scientist at the Naval Surface Warfare Center (Dahlgren, VA); HEUNGSOO KIM is a research scientist and ALBERTO PIQUÉ is a section head in the Materials Science and Technology Division of the Naval Research Laboratory (Washington, D.C.); e-mail: [email protected].