Photoluminescence Spectroscopy: Many explosives share the same peak
Well-known techniques for detecting traces of explosives for security purposes include mass spectrometry and x-ray detection; an active area of research is the use of terahertz radiation (see Laser Focus World, April 2003, p. S10; and November 2004, p. 52). In addition, optical methods exist that are based on fluorescence, photoluminescence (PL), and other phenomena. Researchers at the University of Florida (Gainesville, FL) have discovered that many of the most widely used explosive substances share a common PL peak; the discovery opens the door to a particularly simple PL-based approach.1
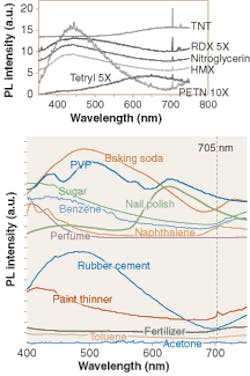
Rolf Hummel and his colleagues tested common explosives, including TNT, nitroglycerin, HMX, PETN, RDX, and Tetryl, and found that, while their long-range PL spectra may vary, they all contained a peak at 705 nm (top). The researchers first used a 25-mW HeCd laser emitting at 325 nm as the excitation source, and a monochromator, fused-silica lenses, and a photomultiplier tube in the detection setup. Some TNT dissolved in a solvent was spread on segments of a clean silicon wafer and dried. Other explosives, also in solution, were deposited on other chips in a similar manner.
Tests on the solvents, as well as on various common nonexplosive substances, showed no 705-nm peak, with the exception of a weak positive result in paint thinner (bottom). Because the explosives contained no taggants, the PL peaks stemmed from the explosives themselves.
The researchers did not confine themselves to the use of a HeCd laser. “The type of laser is not important as long as it emits in the blue to green range,” says Hummel. “We are also using a 532-nm-emitting laser pointer, which is inexpensive. We’ve also used a blue argon-ion laser. All work.” Light-emitting diodes are another possibility, he adds.
The technique is potentially inexpensive, fast, and portable. To simplify the setup, the researchers want to replace the monochromator with two detectors having narrowband optical filters of 690 and 705 nm, respectively. A ratio of the detectors’ outputs provides information on whether the 705-nm peak is present or absent.
The researchers believe the PL peak arises from the existence of two or more nitrogen dioxide groups in the molecular structure of the explosive, either associated with ring structures or otherwise, producing 705-nm emission from interband transitions between certain excited bands and the ground state. The initial setup has a sensitivity in the parts-per-million range, a level that Hummel believes can be improved.
REFERENCE
- To be published in Applied Optics.
About the Author
John Wallace
Senior Technical Editor (1998-2022)
John Wallace was with Laser Focus World for nearly 25 years, retiring in late June 2022. He obtained a bachelor's degree in mechanical engineering and physics at Rutgers University and a master's in optical engineering at the University of Rochester. Before becoming an editor, John worked as an engineer at RCA, Exxon, Eastman Kodak, and GCA Corporation.
