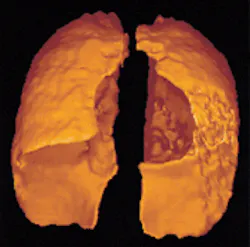
A patient with asthma lies in a magnetic-resonance-imaging (MRI) chamber, taking labored breaths as a doctor viewing a screen watches how the air moves in her lungs. The patient then sits up, takes a shot from her inhaler, and lies down again. The doctor can see immediately how much the inhaler has helped her breathing.
This was one of the tests performed by researchers at the University of Virginia (UVa; Charlottesville, VA), who are studying ways to improve their view into the body's cavities using MRI and hyperpolarized gas. Based on their work, the medical company Nycomed Amersham (Buckinghamshire, England) is preparing clinical trials in an effort to win US Food and Drug Administration approval for this method of imaging.
Magnetic-resonance imaging works by applying a magnetic field to change the spin of protons in water molecules in the bodyµfrom their usual rate of 4 kHz to 63 MHz. But when doctors want to examine a cavity—the inside of the lungs or colon, for instance—they're hampered by the fact that they're looking at empty space. Two harmless gases, however, helium-3 (3He) and xenon-129 (129Xe), have physical properties that make them ideal imaging media (see photo).
The trick is to hyperpolarize the gases, said James R. Brookeman, professor of radiology and biomedical engineering at UVa. Using several gallium aluminum arsenide diode-laser arrays, researchers passed 749-nm beams through a quarter-wave plate and a collimator to circularly polarize them. They trained the light on a glass bulb filled with 3He laced with rubidium vapor. The photons transferred their polarization to the electrons in the rubidium, which has a D1 resonance—a particular quantum-mechanical electrical state—at 749 nm. That, in turn, polarized the 3He. When subjected to the MRI, the polarized protons spin at 48 MHz.
The process is only feasible for 3He and 129Xe because they are noble gases and have the right quantum-mechanical properties. "You can't do this with the regular stuff that you image the body with—the hydrogen atoms in the water of the body," Brookeman said.
It takes several of the laser arrays, made by OptoPower (Tucson, AZ), to produce the 100 W of output needed for the process. Polarization with this method takes several hours, depending on the amount of gas to be treated. "It's just the laser you need," said Brookeman, "but these lasers were not available two or three years ago."
When the group first started, they were only able to polarize about 2% or 3% of a sample of 3He. Now they can polarize approximately 35%, so a patient has to breathe just 300 cc of the gas, instead of the liter that a 3% polarization required. The imaging therefore uses less gas, of which there is a limited supply.
The 3He is a decay product of tritium, used to make triggers for nuclear weapons. With the winding down of the Cold War, the supply of 3He is dwindling. Because 129Xe is a natural component of Earth's atmosphere, researchers would like to use that instead. Brookeman said they have not yet been able to polarize it more than 5%, but that will be the next target of research.
Other applications
Lung imaging is not the only application for hyperpolarized gases and MRI. There has been some preliminary work on imaging the inside of the colon. By examining whether the colon wall is smooth or developing polyps, doctors might be able to predict the onset of colon cancer.
And because 129Xe attaches very easily to the hemoglobin in blood, gas dissolved in the bloodstream could provide other imaging opportunities. The dissolved gas only maintains its polarization for approximately 30 s, but because it only takes approximately 5 s to travel to the brain, there is enough time to get information, Brookeman said.
About the Author
Neil Savage
Associate Editor
Neil Savage was an associate editor for Laser Focus World from 1998 through 2000.