Near-IR Optical Imaging: Shortwave-infrared particles and camera combine for breakthrough in vivo imaging
Compared to other wavelengths, shortwave-infrared (SWIR) light (1000 to 2000 nm) limits autofluorescence, light absorption, and scattering, making biological tissue nearly transparent. For these reasons, SWIR is well suited to in vivo imaging, but a dearth of appropriate imaging agents has gated its adoption for life sciences imaging.
Now, a research group led by scientists at the Massachusetts Institute of Technology (MIT; Cambridge, MA) has developed narrow-range SWIR-emissive indium arsenide (InAs)-based quantum dots.1 The researchers say that the particles provide a dramatically higher emission quantum yield than other SWIR probes, and can be easily modified for various functional-imaging applications.
Their experiments with mice demonstrated important advantages of the dots: deep tissue penetration, high spatial resolution, and multicolor optical imaging. With the help of a state-of-the-art camera, the particles also enabled high-speed image acquisition.
Camera-enabled
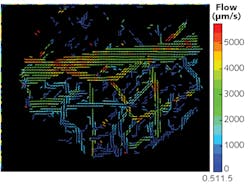
The researchers used the particles—with a commercially available camera featuring a thermoelectrically cooled indium gallium arsenide (InGaAs) focal-plane array (FPA) that is highly sensitive to SWIR light (NIRvana by Princeton Instruments)—to quantify a number of dynamics in awake, unrestrained animals. They measured metabolic turnover of lipoproteins in several organs simultaneously and in real time, as well as heartbeat and breathing rates. Using these readings, they produced detailed, three-dimensional quantitative flow maps of brain vasculature (see figure).
Researchers in the lab of Moungi Bawendi, Lester Wolf Professor of Chemistry, made the dots from semiconductor materials. Tight control of the particles' size and composition enables precise matching of their light emission to desired SWIR frequencies so that they can be easily detected through the surrounding skin and muscle. The particles are "orders of magnitude better than previous materials, and that allows unprecedented detail in biological imaging," according to research scientist Oliver Bruns, who is first author on the paper describing the work. The synthesis of these new particles was first described by Daniel Franke and colleagues.2
Applications
The dots' brightness enables capture using very short exposure times, allows production of video that reveals dynamics such as blood flow, and enables differentiation of veins and arteries. In fact, they are the first particles bright enough to illuminate internal organs in mice that are awake and moving, Bruns says. Not only can the method determine blood-flow direction, it can also track individual blood cells. "We can track the flow in each and every capillary, at super high speed," Bruns adds. "We can get a quantitative measure of flow, and we can do such flow measurements at very high resolution, over large areas."
Initial applications center on preclinical research in animals. "This is an exciting and potentially revolutionary development for small-animal imaging," says Guillermo Tearney, a professor of pathology at Harvard Medical School, who was not involved in this work. "By using probes that are tuned to wavelengths further out in the shortwave near-infrared, the investigators overcome scattering, which is the major phenomenon" limiting such in vivo microscopy, he says.
"In so doing, simpler and less invasive interrogation methods can be utilized to understand structure and function in animal models, on both the organ and cellular level," Tearney says. "I anticipate that these probes will have a major impact on the field of intravital imaging and bioscience research."
Such imaging could potentially be used, for example, to study how the blood-flow pattern in a tumor changes as the tumor develops, which might lead to new ways of monitoring disease progression or responsiveness to a drug treatment. "This could give a good indication of how treatments are working that was not possible before," Bruns says. The researchers are already working on versions using materials that would be safe for humans.
The team included members from Harvard Medical School, the Harvard T.H. Chan School of Public Health, Raytheon Vision Systems, and University Medical Center in Hamburg, Germany, as well as various departments at MIT.
REFERENCES
1. O. T. Bruns et al., Nat. Biomed. Eng., 1, 0056 (2017); doi:10.1038/s41551-017-0056.
2. D. Franke, Nat. Commun., 7, 12749 (2016); doi:10.1038/ncomms12749.
About the Author

Barbara Gefvert
Editor-in-Chief, BioOptics World (2008-2020)
Barbara G. Gefvert has been a science and technology editor and writer since 1987, and served as editor in chief on multiple publications, including Sensors magazine for nearly a decade.