Disease Diagnosis: Miniature spectrometry project aims to improve diagnostics, treatment, and more
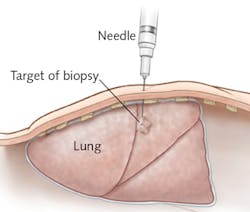
Whether through medical imaging or other means, finding a suspicious lump or mass is alarming. Thanks to image-guided needle biopsy, assessing such abnormalities (by removing tissue or cells for examination by a pathologist) has advanced from a surgical procedure to a minimally invasive one.
But even with image guidance, difficulty in positioning the needle has translated to compromised precision and accuracy. Real-time tissue-characterization feedback at the needle tip has the potential to greatly reduce false negatives and improve the diagnostic results of these procedures, thus advancing cancer treatment and lowering costs. Indeed, several clinical studies have demonstrated the use of photonic needles for spectral analysis of tumors.1-3 But widespread adoption of such systems will require dramatic improvement in cost and size. In particular, the spectrometer console will need considerable development to achieve commercial viability.
The future availability of photonics-needle screening tools at point-of-care locations such as physicians' offices and outpatient centers—along with other implementations of spectroscopic medicine—depends on three things:
- Small, energy-efficient, broadband light sources;
- High sensitivity over a broad spectral range; and
- Fully integrated, high-performance, compact systems.
InSPECT
Integrated Spectrometers for Spectral Tissue Sensing (InSPECT), a three-year European project, launched in 2015 and fully funded with €4,143,460, aims to address these needs (see figure). The project intends to drive spectral sensing into applications that have not been able to access it, enabling its widespread adoption by developing the key photonics building blocks for low-cost miniature spectrometers.
Coordinated in The Netherlands by Philips, the international InSPECT consortium brings together eight partners representing expertise in optics, photonics, device manufacturing, and medical technology: Vrije Universiteit Brussel (Brussels, Belgium), Xenics (Leuven, Belgium), Anteryon (Eindhoven, The Netherlands), Xio Photonics (Enschede, The Netherlands), Aifotec (Meiningen, Germany), Avantes (Apeldoorn, The Netherlands), and the Fraunhofer Institute for Reliability and Microintegration (Germany).
The broadband illumination challenge
Today, illuminating a sample with continuous light covering the visible to near-infrared (NIR) spectrum requires incandescent or halogen lamps that are both bulky and energy-inefficient. InSPECT's answer is to develop a miniature, broadband light source based on wavelength up-conversion of LED emission in photoluminescent materials such as phosphors and quantum dots. The source will emit in the visible (VIS), NIR, and shortwave infrared (SWIR)—that is, 400–1700 nm—and be applied for diffuse reflectance spectroscopy.
The broadband sensitivity challenge
Distinguishing tissues by their molecular fingerprints requires high sensitivity over a broad spectral range (400–1700 nm), which at the short-wavelength end can quickly sample tissue scattering and blood absorption, and at the high wavelength end enables discrimination-for instance, between lipids and water. Currently, this broadband range is possible only through a costly combination of separate spectrometers: InSPECT aims to realize a fully functional, compact, broadband (VIS-NIR-SWIR) spectrometer based on discrete optical components such as wavelength splitters, multiple segments of diffraction gratings optimized for the different bands, and one- or two-dimensional silicon (VIS-NIR) and InGaAs (SWIR) sensors.
The integration challenge
There currently exists no high-performance system that is fully integrated, miniaturized, and cost-effective. InSPECT is working to devise and integrate photonic building blocks for low-cost, miniaturized, tissue-sensing devices. This involves the realization of a miniature low-cost integrated VIS/NIR spectrometer (400–1700 nm), and the InSPECT project has set two paths:
1. The micro-spectrometer, a moderate-risk goal based on the miniaturization and monolithic integration of diffractive dispersive elements and VIS-NIR photodetectors in a small-volume (cubic inch) device, and
2. The nano-spectrometer, a higher-risk goal in which the spectrometer function is realized in a photonic integrated circuit (PIC) based on transparent SiO/SiN waveguide technology using array waveguide gratings (AWG) structures. According to InSPECT coordinator Dr. Jean Schleipen, principal scientist in the Optics Group at Philips, the telecommunications industry previously developed such structures for the SWIR range—InSPECT's challenge is adapting them for the VIS-NIR ranges.
Schleipen says that once the technological challenges are met, the consortium members see many potential applications in biomedicine—and beyond. One example is in implantable devices, where spectral sensing might enable real-time monitoring of (chemo)therapy response. Also, he says, there is a need for miniature spectrometer devices in the food safety and security domains.
During the consortium's biannual meetings, the full team comes together to discuss project planning, resources, and more, including technical achievements. So far, the group has made progress on the light source by synthesizing new fluorescent glasses that can emit NIR-SWIR light when excited by visible LEDs. In addition, an optical design for the microspectrometer is in place and the spectrometer module is currently being assembled—first tests are to be expected in the second half of 2016. They have also designed and manufactured new photonic integration building blocks for the nanospectrometer, and are working on coupling the light from photonic integrated circuits (PICs) into the single-mode PIC waveguides, and on assembling and packaging.
While Schleipen says that InSPECT has not made many public presentations yet, plans have been received well by the general audiences (consortium members have discussed the effort at multiple photonics-focused events, plus the International Society for the Advancement of Cytometry's Cyto conference), and also by physicians that are part of InSPECT's advisory board.
REFERENCE
1. J. Spliethoff et al., Clin. Cancer Res., 22, 2, 1–9 (2016).
2. D. Evers et al., Eur. J. Surg. Oncol., 39, 68–75 (2013).
3. L. de Boer et al., Breast Cancer Res. Treat., 152, 3 (2015).
About the Author

Barbara Gefvert
Editor-in-Chief, BioOptics World (2008-2020)
Barbara G. Gefvert has been a science and technology editor and writer since 1987, and served as editor in chief on multiple publications, including Sensors magazine for nearly a decade.