Quantum-dot research targets general illumination
The ability to generate broad-spectrum white light with good color-rendering qualities is a key step on the road to solid-state lighting for general illumination. One approach, based upon fabricating appropriately distributed combinations of blue, green, and red emitters, can lead to chip designs too costly to compete with conventional fluorescent lighting. Conventional fluorescent lighting is based on the use of phosphors, however, which present an alternative and potentially much more cost-competitive method of generating solid-state white light.
Rather than use a separate source for each primary color, a phosphor composed of three different phosphorescent materials is pumped by a UV source to provide broad-spectrum white light. A difficulty arises in the heat-dissipation-sensitive solid-state-lighting environment, however, because each of the phosphorescent materials has different temperature sensitivity, which can potentially lead to a temperature-sensitive color balance.
In phosphors made from semiconductor nanocrystals or quantum dots, optical properties such as light absorbance and emission wavelengths can be tuned by tailoring the size of the dots, in a single material. Because of the small dot size, most of the atoms are at surface sites, so chemical changes at these sites can also tune the light-emitting properties of the dots. The ability to tune optical properties is also important because LEDs for solid-state lighting typically emit from the near-UV to the blue portion of the spectrum, around 380 to 420 nm. And conventional phosphors used in fluorescent lighting have poor absorption in this range.
Preparation and packaging
The subwavelength dimensions of quantum dots eliminate the light scattering and the associated optical losses that reduce the package efficiency of conventional phosphors by as much as 50%. Nevertheless, nanoparticles tend to have negligible Stokes shift in comparison with conventional phosphors, which leads to low quantum efficiencies.
Researchers at Sandia National Laboratories (Albuquerque, NM) pioneered a synthesis processes for cadmium sulfide (CdS) quantum dots that yielded a quantum efficiency of 76% (in solution) for a blue emission, comparing favorably with 80% to 100% quantum efficiencies obtained with conventional phosphors.1
For quantum dots to be used for lighting, however, they need to be removed from the solution in which they are fabricated and encapsulated (usually in an epoxy or silicone film layer) densely enough to provide bright illumination. Demands on the encapsulation process include that it not alter the surface chemistry of the quantum dots and not allow them to clump up or agglomerate, either of which would alter the optical characteristics. Such a process, also developed at Sandia, achieved 70% quantum efficiency for encapsulated dots.
The encapsulated quantum-dot phosphors at Sandia were formed into a dome over the pump source, a 400‑nm LED chip. The encapsulation process was transferred from Sandia to Lumileds Lighting (San Jose, CA) during the course of a U.S. Department of Energy-sponsored project in which quantum dots incorporated into thin films were evaluated for application to LED chips to produce white-LED lamps based solely on semiconductors.2
Much work yet to do
Lumileds project leader Mike Krames said that when the project ended three years ago the technology, although interesting, was still pretty immature and needed a lot of work prior to direct use within commercial LEDs. “You need high absorption in a thin layer and high density, but the quantum dots interact at high densities,” he said. “So if the quantum efficiency isn’t almost unity, you get a cascading effect, because there is almost no Stokes effect.”
Essentially, the larger dots absorb photons from the smaller ones. So even with 90% quantum efficiency, if the absorption process cascades 10 times, the quantum efficiency effectively drops to about 10%. “The quantum dots exhibit nice properties in aqueous solutions but not in silicon and epoxies,” he added. “They are also strongly affected by temperature in a negative way.”
So while the technology might still prove useful external to the LED, film thicknesses that could provide optical properties of interest for LEDs were too large at the time (on the order of a millimeter), he said. Use in commercial LED packages would require thicknesses on the order of tens of microns, similar to the size of conventional bulk phosphor layers.
Heavy metal
Work toward improving the performance of semiconductor nanocrystal phosphors continues at Sandia. Researchers were unavailable for comment, but published information indicates that approaches have included using nontoxic semiconductor materials to replace the toxic heavy metal cadmium in CdS, and increasing the concentration of quantum dots in the encapsulant to further increase light output.
Applied NanoWorks (Troy, NY) was awarded project funding from the New York State Energy Research & Development Agency (NYSERDA) to work toward improving the levels of brightness obtainable with the company’s existing nontoxic nanophosphors to ultimately serve emerging markets for white LEDs and general illumination.
Applied NanoWorks mass produces nanophosphor products into a bulk powder form, similar in form, cost, and ease of use to commercially available conventional phosphors. The product line is priced in the same ballpark as conventional phosphor materials, has particle sizes on the order of 20 to 30 nm, and is used for research applications that do not require high-brightness illumination.
Tradeoffs involved with moving toward high brightness include size, cost, and the efficiency of the phosphor, according to Applied NanoWorks CTO Partha Dutta. The ideal size for high-lumens-per-watt phosphors is either on the order of 2 nm where scattering effects are minimal or on the order 10 µm with much larger emitters. They are also investigating LED-phosphor design strategies to improve light extraction.
The Solid State Lighting Program in the Lighting Research Center (LRC), directed by Nadarajah Narendran at Rensselaer Polytechnic Institute (RPI; Troy, NY) is working with Evident Technology (Troy, NY), under contract from the NYSERDA, to develop high-quality white-light sources. Rensselaer’s LRC has simulation capabilities to predict efficacies and color properties of white light sources when mixing different wavelength emissions, and Evident can actually create the dots at different sizes to produce the necessary radiation, Narendran said.Direct to white
CEO Rameshwar Bhargava and colleagues at Nanocrystals Technology (Briarcliff Manor, NY) are focusing on a quantum-confined atom (QCA) concept, in which an atom or ion is trapped within a nanocrystal cage, essentially a single-atom doping process that works in reverse of conventional semiconductor technology (see Fig. 1). Instead of the dopant modifying the behavior of the semiconductor, the semiconductor modifies the behavior of the dopant.
The wavelength of light emitted by the trapped atom depends on its chemical properties, while the size of the nanocrystal cage determines the brightness of the emission. When the size of the host cage is reduced from 10 nm to 2 nm, the conversion efficiency of nanophosphors increases rapidly and compares favorably to efficient bulk phosphors.
Current projects at Nanocrystals Technology include a Phase II SBIR grant for bandgap engineering of nanophosphors to improve luminescence characteristics and to develop semiconductor-dopant combinations that will produce white directly when stimulated by blue or UV light.
Last autumn, researchers at Vanderbilt University (Nashville, TN) reported achieving direct blue to white conversion accidentally by using nanophosphors all of the same “magic size.” The CdS crystals all contained either 33 or 34 pairs of atoms, which happens to be a size that the crystals form preferentially, according to Sandra Rosenthal, who leads the research team.3 One of her graduate students came up with an innovative idea to dissolve the nanocrystals in a polymer base so they could be painted onto a blue LED and ultimately yield broad-spectrum white light.
“Our work has triggered interest because it was the first demonstrated single-size nanocrystal to emit white light,” she said. “The main goals going forward are to boost the quantum efficiency and to demonstrate electroluminescence.” Current quantum efficiency is only 3%. But the material has an unusually large Stokes shift. Rosenthal’s team is also using ultrafast spectroscopy and other testing methods to try to determine the mechanism that makes the dots emit white light.
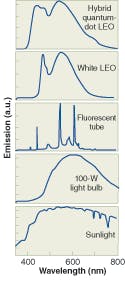
“The light we get almost looks like the sun,” she said. “It’s perfect in that it’s a broad spectrum that cuts off right where the eye stops detecting (see Fig. 2). The color temperature is 5857 K and the appearance is close to that of an incandescent lamp.
“We are cautiously optimistic,” she added. “There are a lot of things to do, such as improving quantum efficiency and making them cost-effectively in large quantities.” The LED that the researchers used to energize the phosphor costs 50 cents, while a white LED costs $5, she said.
REFERENCES
1. www.sandia.gov/news-center/news-releases/2003/elect-semi-sensors/quantum.html
2. www.netl.doe.gov/ssl/rd/LEDSubstrates.html
3. M.J. Bowers II, J.R. McBride, S.J. Rosenthal, J. Am. Chem. Soc. 127(44) 15378 (October 2005).
About the Author
Hassaun A. Jones-Bey
Senior Editor and Freelance Writer
Hassaun A. Jones-Bey was a senior editor and then freelance writer for Laser Focus World.