NANOPHOTONICS: Golden touch proves too hot for optical trapping
Gold nanoparticles can be manipulated with a laser beam more efficiently and sensitively than the glass or polystyrene nanoparticles that serve as handles for laser manipulation of biomolecules. Unfortunately, gold nanoparticles heat up much more than glass or polystyrene as incident laser power increases, to a degree that would damage biomolecules. The efficiency and sensitivity of gold nanoparticles may prove useful in other aspects of biophysical experimentation, however, according to researchers at JILA (Boulder, CO).
The observation, more than a decade ago, that opaque gold nanoparticles could be manipulated with lasers in the same way as the transparent glass or polystyrene beads that are used as handles for experimental manipulation of biomolecules, actually came as a surprise, according to Tom Perkins, leader of the JILA group. But the gold particles used in those initial experiments were too small for practical use. Recent research found that larger gold nanoparticles, of a size that might be used for handling biomolecules, could also be manipulated optically.
But none of the previous research teams looked at heating effects, Perkins said. The experiments were performed at minimal laser power required for trapping. At the higher laser powers required for manipulating biomolecules, however, the metal gets too hot. The measured heating of 266°C/W is 20 times higher than the laser-induced heating of water, rapidly reaching temperatures that could damage biomaterials such as enzymes.
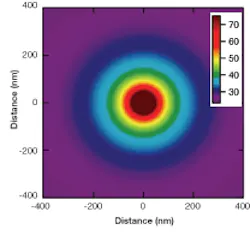
In observing and documenting those heating effects, Perkins’ research team observed inconsistent calibration results from different measurement methods. One method that depended directly on temperature showed an increase that was approximately linear with respect to laser power, but the other two methods that depended directly on water viscosity, which is strongly temperature dependent, showed super-linear increases. The researchers hypothesized that the disagreements were due to local heating of the water and created a model based on that localized heating, which they then verified experimentally (see figure).
The researchers’ optical-trapping instrument used a 1064 nm laser for trapping and 810 and 850 nm diode lasers for position detection, similar to earlier work. Measurements were taken in a buffered aqueous solution 830 nm from the surface by using an estimated laser power at the focus.1
Molecular heaters
Even though the degree of heating is too high for safely handling biomolecules, it can be useful in certain single-biomolecule experiments (melting an RNA “hairpin” for instance), where the idea is to apply heat very locally. Gold nanoparticles might serve as molecular heaters (100 nm area) in such experiments, enabling the “dialing in” of desired temperature with laser power.
For small temperature differences (say up to 10°C), it is generally reasonable to pump infrared light directly into water, which yields about 1°C of temperature increase for every 100 mW at the focus of a 1064 nm laser beam. For experiments in which a local temperature jump from 20°C to 60°C or more is needed, a gold nanoparticle could be used to heat surrounding water in a precisely defined area and in turn precisely stimulate a single biomolecule.
Alternatively, when using a low laser power of less than 1 mW to avoid heating, the enhanced sensitivity of gold nanoparticles offers improved bandwidth and reduced entropic repulsion for zero-force biophysical assays. The researchers also anticipate larger sensitivity enhancements for shorter wavelengths up to a gold particle’s plasmon resonance.
REFERENCE
1. Y. Seol et al., Optics Lett. 31(16) (Aug. 15, 2006).
About the Author
Hassaun A. Jones-Bey
Senior Editor and Freelance Writer
Hassaun A. Jones-Bey was a senior editor and then freelance writer for Laser Focus World.