Nature has spent eons perfecting a host of complex and useful biological substances, some of which can be used by researchers for purposes far removed from the original. In one example, scientists at the University of Cincinnati’s Nanoelectronics Laboratory (Cincinnati, OH) are using salmon DNA to greatly boost the efficiency and brightness of organic light-emitting diodes (OLEDs).1 The resulting devices, called bioLEDs, emit in the green or blue with an order-of-magnitude improvement in performance (while DNA bioLEDs have been fabricated before, the previous versions performed no better than ordinary OLEDs).
The DNA, which was provided by Naoya Ogata of the Chitose Institute (Hokkaido, Japan), was extracted from salmon sperm, a waste product of the salmon-fishing industry. The DNA was first rendered insoluble in water, but soluble in certain organic solvents, and was then reacted with cetyltrimethylammonium (CTMA) to form a DNA-CTMA complex. The DNA-CTMA was then sonically broken up into smaller bits to reduce electrical resistivity. The resulting DNA molecules are about 75 nm long and have a molecular weight of about 145,000 daltons (one dalton is about the mass of a proton or neutron). When situated between hole-injection and hole-transport layers in the OLED, the DNA serves as an electron-blocking layer. Both bioLEDs and conventional OLEDs with common electro-optic-polymer (polymethyl methacrylate, or PMMA; and polyvinyl carbazole, or PVK) electron-blocking layers were fabricated.“The electron-blocking property comes into play when you have a DNA thin film (and these films are of the order of 10 to 20 nm thick) sandwiched between other organic layers; in other words, when you try to transport electrons across the DNA film, not along it,” says Andrew Steckl, one of the researchers. “The electron blocking is a consequence of the difference in the vacuum levels of the DNA (small) and the material next to it that supplies or transports the electrons.”
The blue and green bioLEDS, which were 2 mm square, were compared with similar-size conventional organic LEDs (OLEDs) in an experiment (see Fig. 1). At a current density of 200 mA/cm2, the green-emitting bioLED was more than three times as bright as the control OLED (15,000 versus 4500 cd/m2); at the same current density, the blue bioLED was almost twice as bright as the control (1500 versus 800 cd/m2). When pushed to a current density of 310 mA/cm2, the green bioLED reached a luminance of 21,000 cd/m2.
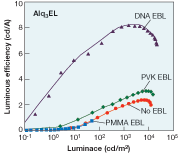
Luminous efficiency of the bioLEDs was substantially higher than that for the control OLEDs (see Fig. 2), peaking at 8 cd/A, compared to a maximum of 3 cd/A for the best runner-up.
“The modified DNA is surprisingly robust,” says Steckl. “Our preliminary results show that the lifetime of the bioLEDs are significantly longer than that of equivalent OLEDs without the DNA layer. We are working on understanding the difference in degradation mechanisms.”
REFERENCE
1. J. A Hagen et al., Applied Physics Letters 88, 171109 (April 24, 2006).
About the Author
John Wallace
Senior Technical Editor (1998-2022)
John Wallace was with Laser Focus World for nearly 25 years, retiring in late June 2022. He obtained a bachelor's degree in mechanical engineering and physics at Rutgers University and a master's in optical engineering at the University of Rochester. Before becoming an editor, John worked as an engineer at RCA, Exxon, Eastman Kodak, and GCA Corporation.