UV spectroscopy becomes a powerhouse for emissions monitoring
Danfoss IXA, a Danish multinational company, develops marine-emission analyzers based on ultraviolet (UV) absorption spectroscopy to monitor the nitrogen oxide (NOx), sulfur dioxide (SO2), and ammonia (NH3) emissions released by cargo ships. An optical device is located inside the exhaust systems of the ships, exposing the system to a harsh environment with extreme temperatures, vibrations, and a corrosive environment, which leads to unique environmental requirements for the spectroscopy system. It allows the shipping vessels to ensure they are complying with all environmental regulations.
Why monitor smokestack emissions?
Maritime smokestack emissions from international shipping vessels contribute to premature deaths of people around the world from lung damage and cardiovascular diseases (see Fig. 1). The global annual cardiopulmonary and lung cancer deaths attributed to shipping emissions are estimated to be as high as 60,000.1 As seen in Figure 2, this is more than the estimated annual deaths from both natural disasters and heat/cold exposure combined. It’s a severe problem that impacts human health and also damages ecosystems in the ocean and on land.
The International Maritime Organization (IMO) and U.S. Environmental Protection Agency (EPA) established Emissions Controlled Areas (ECAs) in the oceans around many countries, with strict emission regulations—it’s impossible to access many major ports without sailing through them.
Without analyzers like the ones developed by Danfoss IXA, for example, there are no other convenient and reliable ways for the authorities to monitor ships’ emissions and enforce these regulations. While many local and regional initiatives designed to limit the air emission from ships exist, enforcing these policies is difficult. Spectroscopy-based marine emissions analyzers are powerful tools to accurately measure a ship’s air emissions in real time.
UV spectroscopy system
The fundamental principle of spectroscopy is that substances have unique absorption spectra, absorbing different wavelengths of light based on their atomic and molecular composition. Danfoss IXA’s UV spectroscopy system consists of a high-intensity UV light source, a UV spectrometer, and UV enhanced optical components such as fibers, a lens, and a mirror (see Fig. 3). To see how different wavelengths are absorbed to determine the composition of exhaust fumes, the spectrometer spatially separates the broadband emissions from the light source onto a 1D detector array, which simultaneously measures the whole UV spectrum.
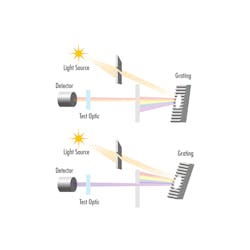
While Danfoss IXA’s system does not use a monochromator for wavelength isolation, many spectroscopy systems do. In these cases, light from the UV source enters the monochromator’s entrance slit where it splits into its component wavelengths using a dispersive element, such as a diffraction grating or prism (see Fig. 4).
The monochromator’s exit slit then blocks all wavelengths except for a narrow band that passes through the sample of exhaust moving through the device. Changing the angle of the diffraction grating or prism changes the wavelengths that pass through the exit slit, which allows the test wavelength band to be finely tuned. Light passing through the sample is then directed onto a detector to determine the absorption that occurred; it is then used to calculate the molecular composition of the exhaust.
For monochromators that use diffraction gratings, the grating’s groove frequency is typically measured as the number of grooves per millimeter. Higher groove frequencies improve optical resolution, but result in a narrower usable wavelength range. On the other hand, lower groove frequencies lead to a broader usable wavelength range—but with a sacrifice of resolution.
Environmental requirements
The development of such a system is very challenging because of the extreme high temperature and pressure requirements. High temperatures can cause optics to fail due to melting and thermal stresses, which severely limits the types of optical materials that can be used. High temperatures can also cause adhesives in the optical assembly to outgas and contaminate the system. This stem is exposed to temperatures as high as 500°C, so its high pressure requirements make the sealing of the optical system critically important. The optics need to transmit UV light with little absorption, which also limits the available optical materials.
UV degradation of optics
Another challenge this project faces is that UV optics tend to have limited lifetimes, largely due to contamination arising from the high-power UV photons interacting with the environment and UV light breaking down the coatings and substrates of the optics. Both effects degrade the performance of optical components over time.
Unwanted material can be deposited on the surface of the optics when high-power UV light interacts with particulates, water vapor, organics, and other contaminants in a system. Outgassing and other airborne molecular contaminants commonly lead to carbon-based deposition on optical surfaces. An example of the dendritic growth of UV-caused contamination is shown in Figure 5.
Interactions with gases surrounding the optics also lead to the deposition of contaminants, so any exhaust entering the system is a source of contamination. The photon energies of UV wavelengths less than 400 nm approach the same scale as bond energies of the surrounding molecules, which allows UV light to break down some of these bonds. This then creates other ions and molecules that can contaminate optical surfaces.
The coating and substrate materials of UV optics themselves are also susceptible to degradation over time with high-power UV light exposure, thanks to the process of optical fatigue. Numerous uses over time cause them to break down, and it results in discoloration or other changes in the material. Their refractive index can be modified to produce a lensing effect that can increase the local intensity. Self-trapped excitons can also be formed, which leads to the accumulation of absorption centers.
Because of these effects, UV optics may need to be replaced over time—but proper sealing, purging, and cleaning mitigate these effects.
The demanding environment of Danfoss IXA’s gas emission analyzers introduced many challenges to the system’s optical and optomechanical design, but the device proved successful and is now helping monitor the emissions of thousands of shipping vessels across the globe.
This is a great victory for the environment—a step toward minimizing the NOx, SO2, and NH3 emissions of international shipping. Any reduction in this pollution helps reduce the number of annual cardiopulmonary and lung cancer deaths attributed to shipping emissions.
When designing optical systems operating in demanding environments, discuss the specific environmental requirements with your optical component manufacturer. They should be able to guide you through the key considerations, clearly explain any tradeoffs that may need to be made, and ensure your system performs as needed.
REFERENCES
1. M. Dwortzan, “Smarter regulation of global shipping emissions could improve air quality and health outcomes,” MIT News (Aug. 17, 2021); https://tinyurl.com/yt4sxr2a.
2. H. Ritchie, F. Spooner, and M. Roser, “Causes of death,” Our World in Data (Dec. 2019); https://ourworldindata.org/causes-of-death.
3. B. Arnold, C. Rashvand, L. Willis, and M. Dabney, Proc. SPIE, 12300 (Dec. 2, 2022); doi:10.1117/12.2638404.
About the Author
Cory Boone
Cory Boone is technical marketing manager at Edmund Optics. He is responsible for managing the creation of technical marketing content, including application notes, published articles, web copy, email campaigns, video scripts, trade show materials, case studies, and other marketing literature. He plans the company’s technical content strategy and acts as the technical voice of the marketing department. Cory is also active in educational outreach and spreading general awareness of the optics industry. He received a B.S. in Optical Sciences and Engineering along with minors in Math and Material Science Engineering from the University of Arizona.
Samara Gomes
Marketing Coordinator, Danfoss IXA
Samara Gomes is marketing coordinator at Danfoss IXA (Kolding, South Denmark, Denmark).

![FIGURE 2. 2019 global causes of death, showing that deaths attributed to shipping emissions outnumbered those from both natural disasters and heat/cold exposure combined. [1, 2] FIGURE 2. 2019 global causes of death, showing that deaths attributed to shipping emissions outnumbered those from both natural disasters and heat/cold exposure combined. [1, 2]](https://img.laserfocusworld.com/files/base/ebm/lfw/image/2023/09/2310LFW_cb_2.64fb53cd2e55e.png?auto=format,compress&fit=max&q=45?w=250&width=250)

![FIGURE 5. Example of UV-exposure-induced contamination on an uncoated fused silica window. However, this image was captured after 6 weeks of ~3 W UV laser illumination, which is a different use case than Danfoss IXA’s gas analyzer, but it serves to illustrate the type of UV contamination that can occur. [3] FIGURE 5. Example of UV-exposure-induced contamination on an uncoated fused silica window. However, this image was captured after 6 weeks of ~3 W UV laser illumination, which is a different use case than Danfoss IXA’s gas analyzer, but it serves to illustrate the type of UV contamination that can occur. [3]](https://img.laserfocusworld.com/files/base/ebm/lfw/image/2023/09/2310LFW_cb_5.64fb53cdd1c79.png?auto=format,compress&fit=max&q=45?w=250&width=250)
