Anticancer therapeutic agent bombs cancer cells
A new anticancer therapeutic agent, inspired by marine animals, can very specifically target cancer cells. Called a bomb-like agent, it essentially detonates cancer cells, and only those cells.
According to a team in South Korea—led by researchers at Pohang University of Science and Technology (POSTECH), in conjunction with researchers from Kyungpook National University—cancer treatment typically comprises several therapies (rather than a single type of treatment), as the disease takes multiple biological pathways in the body. Dispensing multiple therapeutic agents simultaneously to very specific sites of the cancer can be difficult, however.
The team has developed a way around this challenge, with photoactivatable proteinic nanoparticles (NPs) “capable of allowing biological photoabsorption and near-infrared (near-IR) responsive anticancer therapeutic effects.”
The new agent generates heat in precise areas where near-IR laser light is illuminated, generating nitric oxide gas (providing an anticancer effect). It then “simultaneously releases anticancer drugs to achieve the trimodal treatment of photothermal-gas-chemotherapy.” According to the study, published in Advanced Healthcare Materials, the incorporation of a thermo-sensitive nitric oxide donor and doxorubicin (a drug used to treat cancer) “leads to synergistic anticancer therapeutic effects as a result of photothermal-triggered ‘bomb-like’ multimodal actions.”
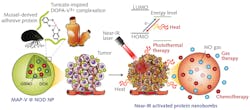
Specifically, this new therapeutic agent is a photoactivated—in which properties change depending on the external light—“nanobomb” featuring mussel adhesive proteins (MAPs) as well as the catechol-vanadium complex of tunicates (marine invertebrate animals that are typically found attached to the underside of a boat or under docks; see figure) “in light-related energy conversion through electron transfer.”
The NPs “retained on cancer cells for a prolonged period contributed by the strong adhesiveness of MAP,” according to the researchers, who note that “in addition, great biocompatibility and biodegradability of proteinic nanoparticles resolve the safety issue of conventional light-responsive nanotherapeutics.”
As nitric oxide gas biometabolizes quickly, it can be difficult for it to retain its anticancer effects. However, the researchers say the nanobomb can control the gas release with light, “which greatly increases the delivery efficiency” of the gas.
“This nanobomb can be applied not only to photothermal-gas-chemo-therapy, but also to the delivery of therapeutics including genes and antibodies or contrast agents,” says POSTECH Professor Hyung Joon Cha. “And therefore [it] can be widely used according to the specific needs of the patient or the condition.”
In the study, NPs were created by adding the catechol-vanadium complexes—which help light and electrons move in tunicates—to the MAP. When these NPs are irradiated with near-IR lasers, the temperature can be raised to 50°C within five minutes, prompting photothermal conversion efficiency to about 50%.
The study has included preclinical trials with animal models, in which an individual tumor redeveloped 15 days after being treated with a single photothemal treatment. When the researchers administered the trimodal treatments with the nanobombs, they saw prolonged effectiveness of the therapy—for about 30 days, the team saw no tumors.
“With a single nanoparticle, various site-specific therapeutic agents can be locally administered and multimodal treatments can be easily controlled with a single stimulus,” Cha says, “which will be effective in treating cancer patients in the future.”
About the Author
Justine Murphy
Multimedia Director, Digital Infrastructure
Justine Murphy is the multimedia director for Endeavor Business Media's Digital Infrastructure Group. She is a multiple award-winning writer and editor with more 20 years of experience in newspaper publishing as well as public relations, marketing, and communications. For nearly 10 years, she has covered all facets of the optics and photonics industry as an editor, writer, web news anchor, and podcast host for an internationally reaching magazine publishing company. Her work has earned accolades from the New England Press Association as well as the SIIA/Jesse H. Neal Awards. She received a B.A. from the Massachusetts College of Liberal Arts.