Holographic optical tweezers manipulate nanobiorobots
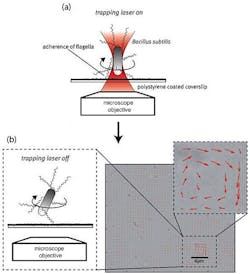
IMAGE: A schematic shows the (a)adhesion process of a self-propelling bacterium on a coated surface based on a holographic optical tweezer (HOT). A bright-field image shows multiple B. subtilis attached to a coated surface (b) and red arrows superimposed on the image indicate the flow of the surrounding fluid resulting from bacterial actuation. (Image credit: SPIE)
Different approaches for exploiting the motion of bacterial nanomotors have been used in the last few years. For instance, self-propelling bacteria, such as Escherichia coli and Bacillus subtilis, attached on solid surfaces (bacterial carpets), have been used to pump liquid and to enhance mixing in microfluidic systems. In addition, they have been used to transport or rotate microparticles, ranging from conventional microspheres to more complex abiotic objects such as microscopic gears.
RELATED ARTICLE: Plasmonic nanostructures are optimized for optical trapping
A prerequisite for reasonable use of such biological motors in general and efficient co-operative behavior in particular is an attaching method (that is, to adhere or tether a single or a multitude of individual biological cells to a specific surface or microparticle in a defined way) that gives the best possible control over the location and state of every single cell. However, micropatterning and microassembly processes require relatively high effort, suitable material know-how, and equipment. Moreover, fine control over individual bacteria is very limited, and the approach lacks flexibility and re-configurability with respect to new arrangements.
Instead, researchers Cornelia Denz, Alvaro Barroso, Florian Hörner, and Shirin Landwerth of the Institute of Applied Physics (IAP) at the University of Münster (Münster, Germany) have developed a general solution to this problem that uses a holographic optical tweezer (HOT) workstation that can simultaneously create tens of optical traps to control the assembly of single bacteria at specific locations.
To achieve these optical trapping landscapes, the team modulates the phase front of a laser beam with a phase-only spatial light modulator (SLM) to generate multiple optical tweezers at the sample plane. Each of the optical traps can be controlled independently in real time so that several bacterial cells and particles can be manipulated simultaneously. In addition, suitable selection of the laser wavelength (here, 1064 nm), power level, and exposure time ensures that light absorption by the biological specimen is insufficient for serious damage during assembly.
By combining the HOT's full 3D manipulation of multiple bacteria with controlled attachment on functionalized surfaces, they have demonstrated actuation of microsystems by self-propelling bacteria. In the first scheme, they optically trapped, moved, and attached arrays of multiple bacterial cells in a controlled way on homogeneous and inexpensive polystyrene-coated surfaces (see figure). After applying the trapping laser beam, the bacteria rotated, typically at frequencies of a few hertz, around a rotation axis in close proximity to the cell body as indicated by their relative positions in the image sequence. These bacteria are not only obviously motile, but following a simple mechanical model are tethered to the surface with exactly one flagellum. In this way, rotation of the bacterial microsystem induces a continuous flow that could mix fluids or induce dynamic fluid motion.
The team further applied the HOT's ability to dynamically control and arrange tens of nonspherical particles in any desired configuration to assemble biohybrid microrobots consisting of a single self-propelling bacterium and a single payload particle. The propulsion of the optically assembled microrobot shows the potential of the proposed method as a powerful strategy for fabricating biohybrid microrobots, which could be envisioned as transport vehicles for drug delivery to otherwise inaccessible places.
Moving forward, the group plans to use functionalized nanocontainers with biomolecules that are known for interaction with cell surfaces. This will open up future possibilities for fabricating biomedical microrobotic systems.
SOURCE: SPIE Online; http://spie.org/x114975.xml?highlight=x2416&WT.mc_id=KNRBIOMEDE