Adaptive optics sharpens multiphoton-microscope images
Originally developed for astronomy and military use, adaptive-optics is coming into its own for use in optical-sectioning microscopy techniques such as multiphoton microscopy.
Optical microscopy is a fundamental tool for life-sciences research, providing submicron-resolution images of cellular structure. In the last 20 years, with the wide availability of laser-scanned confocal microscopes, the ability to visualize structure in three dimensions without having to physically slice the sample has further added to the importance of the technique.
With the development of multiphoton microscopy in 1990, the capability to image with potentially high resolution deeply within live samples has opened up new possibilities for research and long-term clinical diagnosis based upon the examination of micron-sized features within tissue.1 The requirements of the life scientist in this area have lead to significant developments in optical physics with high-performance hands-free femtosecond lasers, improved detectors, and high-performance computing. But there are still limitations imposed on such optical sectioning techniques as one moves deeper into the sample. Research is now under way in the U.K. to overcome these limits by using adaptive-optics technology originally developed for ground-based astronomy and military observations.
The importance of a good PSF
Fundamental to all laser-scanning microscopy is the size of the focused spot within the sample. Microscope-objective manufacturers go to extreme lengths to produce diffraction-limited point-spread functions (PSFs). In the design process, however, assumptions have to be made about the conditions under which the objective will be used. For sensible reasons, the assumption is made that the sample has a uniform refractive index either of air, water, or oil (matched to the microscope slide). When imaging single thin cells, these are valid assumptions, but as one moves deeper into the sample the optical properties of the tissue start to distort the incoming and returned wavefronts, leading to a significant loss of resolution and image quality as one moves into the sample.
With the ability of multiphoton microscopes to image at depths of up to 2 mm, the sample-induced aberrations begin to play a limiting role in the imaging performance of the instrument. An additional factor must be considered for multiphoton microscopes because the process depends on the nonlinear excitation of the fluorophores within the sample. As the PSF degrades, the intensity of the laser spot falls, with an associated increase in excitation volume and loss of signal. The signal can, in principle, be recovered by increasing the laser power, but only to the point at which the sample can begin to be damaged.
The increase in PSF size caused by tissue-induced aberrations is analogous to the disturbance of images from ground-based telescopes caused by atmospheric turbulence. Although the structures of biological specimens are typically static, variations in the induced aberrations occur as the focused beam is scanned across the specimen. Adaptive-optics technology developed for astronomy can be applied to reduce the aberrations in confocal and multiphoton microscopes imaging at depth within a sample. In a multidisciplinary project, two groups have been looking at this problem with alternative approaches to solving the challenge, both based around the use of flexible-membrane mirrors as the active optical element.
Measuring aberrations of real samples
Although samples of tissue have been examined microscopically for hundreds of years, nobody had previously measured the wavefront disturbance produced by tissue samples. Researchers in the Engineering Department at the University of Oxford (Oxford, England) have developed a method of measuring the aberrations from a selection of biologically interesting samples.2 Based upon phase-stepping interferometry, the complex phase distortions introduced by samples up to 100 µm thick have been recorded.
In the setup, a 633-nm beam from a HeNe laser is split into measurement and reference beams. The measurement path includes a 40×, 0.6-numerical-aperture (NA) condenser lens that focuses the light into the specimen, which is examined in transmission. Objective-lens choices include a 20×, 0.8-NA oil-immersion lens and a 20×, 0.5-NA dry lens. The phase-stepper in the reference path is a piezo-translated mirror. A CCD image sensor is conjugate to the pupil plane of the objective lens. Phase-stepping is synchronized at 25 Hz with the camera for video-rate measurement. (The measurement-optics path is switchable to an ordinary brightfield configuration for preliminary examination of the specimen.)
Spherical aberrations predominated in the specimens, ranging up to approximately 0.5 Zernike units, with other field-dependent modes contributing. The results reveal how biological specimens affect the imaging properties of microscopes, allowing the researchers to quantify the various effects of scattering and image distortion as well as aberrations (see Fig. 1). The results show that low-cost adaptive optics can provide considerable improvements in imaging quality because the compensation of aberrations (both spherical and field-dependent) moves the performance toward the diffraction limit.
Putting adaptive optics to use
Linked with this approach is the use of the adaptive-optic element to act as a wavefront sensor and corrector in a complete imaging system.3 In a version of this technique called modal wavefront sensing, a sequence of predetermined distortions (as a series of Zernike aberration terms) is applied to a deformable mirror and the resulting focal intensities are measured. The amplitude of each Zernike aberration mode is then obtained using a simple calculation. The method can also use the fluorescence signal from a confocal or multiphoton microscope and has been shown to improve image quality.
An alternative approach to the problem is being developed at the Institute of Photonics at the University of Strathclyde (Glasgow, Scotland). In this work, the actual distortion induced by the sample is not measured; instead, the actual image is used as the basis for an optimization algorithm.4 After passing through an x/y raster-scanning system, the beam is directed onto a membrane mirror (fabricated by Flexible Optical B.V. of Delft, Netherlands) via a polarizing beamsplitter and quarter-wave plate. The light is reflected from the mirror and through a piezo-controlled objective lens (see Fig. 2). When the system is used in the multiphoton configuration, the emission light is detected directly after the objective, whereas in the confocal mode the light is again reflected off the active mirror before being descanned and subsequently detected. The optimization of the mirror is controlled using several optimization routines using either image contrast or brightness in a region or the entire image as selected by the user.
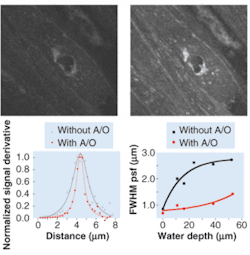
FIGURE 2. A multiphoton image shows a 32 × 32‑µm x-y slice of a guinea-pig bladder at a depth of 30.8 µm into the sample. An unoptimized image lacks detail (top left); a similar image benefits from the use of adaptive optics to compensate for tissue-induced aberrations (top right). The multiphoton point-spread function (PSF) at an 11-µm water depth with and without adaptive optics (A/O) shows the improvement resulting from A/O (bottom left). A plot of PSF full width at half maximum (FWHM) as a function of depth for a 1.4-numerical-aperture 40× oil objective illustrates both the increase of FWHM with water depth and the improvement possible with the use of A/O (bottom right).
In the initial work the main focus was on the use of a conventional hill-climbing algorithm, but, although fast, the system does not always reach the global maximum. More recently, the group has used a wide range of genetic and simulated-annealing routines that statistically offer a better solution, but at the expense of time. The results to date indicate that significant improvements in image quality are possible in both confocal and multiphoton microscopy. The user can select the area of the image that is of most interest to the life scientist and then optimize the mirror to enhance the features of interest.
ACKNOWLEDGMENT
The work described here forms part of a project funded by the Research Council U.K. Basic Technology Programme and was presented at the Optical Society of America’s 2004 Annual Meeting in Rochester, NY.
REFERENCES
1. W. Denk et al., Science 248, 73 (1990).
2. M. Schwertner et al., J. Microsc. 213, Pt. 1, 11 (January 2004).
3. M. J. Booth et al., Proc. Nat. Acad. Sci. 99(9) 5788 (April 30, 2002).
4. P. N. Marsh et al., Optics Express 11, 1123 (2003).
JOHN GIRKINis associate director of the Institute of Photonics, University of Strathclyde, 106 Rottenrow, Glasgow G4 0NW, Scotland; e-mail: [email protected]. MARTIN BOOTH is a Royal Academy of Engineering/EPSRC research fellow at the Department of Engineering Science, University of Oxford, Parks Road, Oxford OX1 3PJ, England.