Near-field microscopy moves into the mainstream
scanning OPTICAL MICROSCOPES
Near-field microscopy moves into the mainstream
George J. Collins
Lens-free scanning optical microscope sidesteps the diffraction limit to image with nanometer-level resolution.
Nearly 500 years have passed since the invention of the optical microscope. While the capabilities of the instrument have been greatly extended by improved lenses, new contrast mechanisms, and the use of lasers, the magnification power of the optical microscope has remained essentially the same. Diffraction inherent in all magnifying lenses limits the best possible resolution and maximum usable magnification to approximately one-half the wavelength of light. For blue light this resolution limit is about 200 nm, but, in practice, the resolution limit for optical microscopes is closer to 500 nm.
Observation of features on the order of 500 nm has allowed scientists to make dramatic advances in science and technology. Understanding bacteria and cells has revolutionized biology and medicine. The inability of optical microscopes to resolve features smaller than the resolution limit, however, delayed the discovery and study of viruses and important nanometer-size cellular components such as DNA.
Microscopy has other applications in addition to the passive observation of specimen structure in medicine and biology. Imaging optics can be used in an active mode to pattern submicron structures onto specimens, for example in optical lithography. One of the goals in optical lithography is reduction of feature size. The smaller the features, the more circuit elements can be packed onto a chip, increasing computing power and storage space. Once again, the diffraction barrier is the limiting factor, preventing the fabrication of features much smaller than 0.25 µm. New technologies must be developed to permit fabrication of ever smaller features in future-generation computer chips and ultrahigh-capacity optical-data-storage media.
Development of near-field microscopy
In a 1928 discussion with Albert Einstein, E. H. Synge proposed a novel optical microscope that had no lenses in the part of the microscope that defines resolution.1 In theory, this "near-field" microscope could overcome the 500-year-old diffraction barrier that prevented higher optical resolution. The proposed instrument would illuminate the specimen under inspection through a tiny aperture, scanning the spot of light across the sample. If the aperture were smaller than the wavelength of the illumination and were held within a few nanometers of the sample (in the near field), the resolution limit of this microscope would be primarily defined by the diameter of the aperture. Unfortunately, the technology needed to build a near-field optical microscope did not exist in 1928.
With the invention of the scanning tunneling microscope (STM) in 1982, a mechanism for scanning a sharp probe within a few nanometers of the surface of a sample became available.2 In 1984, Dieter Pohl of IBM (Zurich, Switzerland) demonstrated the use of a metal-coated glass fiber in an STM-like microscope, producing the first working version of the near-field scanning optical microscope (NSOM) that Synge had proposed half a century earlier.3 IBM received a patent for this optical near-field scanning microscope.
Between 1984 and 1991, a number of researchers built near-field optical microscopes, but optical transmission efficiencies on the order of 10-7 to 10-8 for the glass fiber and pipette probes limited the analytical usefulness of the instruments.
In 1991, Eric Betzig and Jay Trautman of AT&T Bell Laboratories (Murray Hill, NJ) invented and patented an adiabatically pulled fiberoptic probe. Unlike glass pipettes, the fiber probe features a high-refractive-index (RI) core that confines and refocuses the light as it passes down the fiber. High-RI core probes have achieved 100 to 10,000 times the optical transmission of earlier probe designs that included glass fiber or glass pipette probes with low-RI air cores.4 Combined with shear force feedback, a newly developed feedback mechanism that accurately maintained tip to sample separation, the high optical throughput of the adiabatically pulled probes allowed a significant amount of analytically useful work to be done with the NSOM.5 AT&T Bell Laboratories also received a patent for shear force feedback.
In 1992, Betzig and Trautman published a landmark paper demonstrating super-resolved transmission, reflection, polarization, refractive index, and fluorescence near-field scanning optical microscopy.6 (Super-resolved describes resolution better than the l/2 Rayleigh diffraction limit. Resolution of l/20 has been achieved by near-field scanning optical microscopy.) They also demonstrated the ability to do super-resolved optical spectroscopy. Betzig subsequently demonstrated detection of single fluorescent molecules using near-field scanning optical microscopy, and Trautman showed optical spectra of individual fluorescent molecules.7,8
Early in 1994, TopoMetrix introduced the Aurora, the first commercial NSOM (see Laser Focus World, Oct. 1993, p. 105). Now, this system is capable of super-resolved transmission, reflection, polarization, refractive index, and fluorescence imaging. Super-resolved optical spectroscopy and single molecule detection have also been demonstrated with the Aurora.9,10 In addition, it has performed infrared near-field scanning optical microscopy, Raman near-field scanning optical microscopy, near-field scanning optical microscopy in liquids, and gigahertz electro-optic sensing.11 New applications are continuing to emerge.
Types of near-field optical microscopy
To demonstrate super-resolved transmission imaging, a glass slide was coated with a pattern of 60-80-nm-diameter platinum carbide (PtC) dots (see Fig. 1). In the transmission NSOM image, the PtC dots block the light and appear as dark spots 60 to 80 nm wide. In reflection mode, the metal dots would reflect light coming from the probe and appear as bright spots on a dark background.
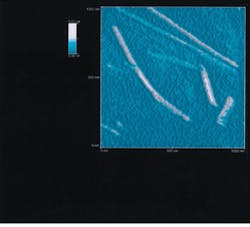
Tobacco mosaic virus, an 18-nm-diameter rod-shaped virus, was mounted on mica and scanned by a transmission NSOM (see Fig. 2). Although this virus is 27 times smaller than the 488-nm light emanating from the aperture of the probe, the microscope images it clearly.
In addition to passive imaging, the NSOM can be used in an active mode to perform subwavelength surface modification, for example in a 300-nm-thick liquid-crystal film (see Fig. 3). The highly oriented molecular structure of the liquid crystal polarizes reflected light. A polarizing filter placed in front of the detector was oriented to transmit polarized light, producing the bright background in the image.
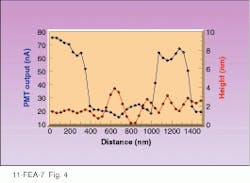
The rectangular dark areas were previously scanned by the NSOM probe with enough power to change the orientation of the liquid-crystal molecules. This changed the polarization of light reflected from those regions, causing it to be blocked by the polarizer in front of the detector and making those areas of the image dark. Line profiles and topographic images of the film show that the liquid crystal is topographically flat to within ۬ nm, yet the optical image switches from bright to dark (see Fig. 4). This shows that optical contrast is independent of topography. It is important to note that this switch from bright to dark takes place within 60 nm, demonstrating the potential for subwavelength optical lithography.
Fluorescence imaging
A variety of compounds are used as sensitizers for photographic film. These materials are coated onto the surface of silver halide grains to extend the range of lighting conditions that will "expose" the film by reducing silver halide to silver. J-aggregates of 1,1`-diethyl-2,2`-cyanine iodide are used for this purpose. Understanding the structure and optical properties of j-aggregates can help improve their use as sensitizers in fine-grained photographic films.
D. Higgins and P. Barbara (University of Minnesota, Minneapolis, MN) produced fluorescence NSOM image j-aggregates, revealing long, thread-like structures suspended in polyvinyl sulfate and spin-coated on glass. To obtain these images, the specimens were illuminated with 514-nm light through the NSOM fiber, and fluorescence in the 540- to 640-nm range was passed to the detector, imaging j-aggregate structures with 65- to 70-nm resolution.
Single-molecule detection demonstrates the sensitivity of NSOM fluorescence imaging (see photo on p. 104). The molecules of diI (1,1`-didodecyl-3,3,3`,3`-tetramethyl indocarbocyanine perchlorate) were spread in a submonolayer and imaged. (A submonolayer is formed when a very dilute solution is spread on a surface and the solvent evaporates. The nonvolatile solute molecules lie on the surface in a widely spaced pattern. A monolayer would be complete coverage one molecule thick.) The ability to detect single fluorophores could be important in high-resolution biological applications.
Near-field scanning optical microscopy was moved from theory to practice by the scanning probe microscope, the high-RI-core fiber probe, and shear force feedback. In the few short years following these inventions, a wide variety of NSOM imaging and analytical capabilities have been demonstrated and are now used in real-world applications. Proven, accepted, and available, near-field scanning optical microscopy will play an important role in analytical microscopy in the 1990s. n
REFERENCES
1. E. H. Synge, Phil. Mag. 6, 356 (1928).
2. G. Binning et al., Phys. Rev. Lett. 49, 57 (1982).
3. D. W. Pohl, W. Denk, and M. Lanz, Appl. Phys. Lett. 44, 651 (1984).
4. T. D. Harris, R. D. Grober et al., Appl. Spectroscopy 48, 1, 14A (1994).
5. E. Betzig, P. L. Finn, and J. S. Weiner, Appl. Phys. Lett. 60(20), 2484 (1992).
6. E. Betzig and J. Trautman, Science 257, 189 (1992).
7. E. Betzig and R. J. Chichester, Science 262, 1422 (1993)
8. J. K. Trautman, S. S. Macklin, and E. Betzig, Nature, 369, 40 (1995).
9. D. Higgins and P. Barbara, J. Phys. Chem. 99(1), 3 (1995).
10. D. Higgins and P. Barbara, oral presentation at Scanning 1995, Monterey, CA.
11. C. Böhm et al., J. of Phys. D, (1994).
Near-field scanning
optical microscope image shows detection of single molecules of the fluorescent dye 1,1` didodecyl-3,3,3`,3`-tetramethyl indocyanine perchlorate. The variation in peak shapes and intensities can be related to the orientation of individaul dye molecules on the surface of the microscope slide.
FIGURE 1. Transmission near-field scanning optical microscope images of 60-80-nm-diameter platinum carbide (PtC) dots on a glass substrate (top) show regions of bare glass not revealed in topographic image (bottom). The opaque PtC dots form the hexagonal patterns of dark spots seen in the image.
FIGURE 2. Transmission near-field scanning optical microscope image of 18-nm-diameter, rod-shaped tobacco-mosaic-virus particles demonstrates the resolution achievable with this technology.
FIGURE 3. Cross-polarization, reflection-mode near-field scanning-optical-microscope image of a liquid crystal shows rectangular areas where "lithography" was performed with a near-field scanning-optical microscope.
FIGURE 4. Plot of PMT output shows that near-field scanning-optical-microscope-written liquid-crystal film changes from light to dark over 60 nm while remaining topographically flat.