NONLINEAR OPTICAL MICROSCOPY: Ultrafast SPOPO enables deeper tissue imaging
GAIL McCONNELL
Nonlinear-microscopy techniques such as second- and third-harmonic generation (SHG and THG) and multiphoton laser-scanning fluorescence microscopy (MPLSM) are very useful for deep imaging of biological tissue; their capabilities, however, are entirely dependent upon the availability of suitable laser technology. The current industrial-standard excitation source for nonlinear microscopy is the femtosecond-pulse Ti:sapphire laser. Although this powerful source can provide more than 100 mW average power at the sample with around 80 MHz repetition rate, its wavelength range is limited by the bandwidth of the Ti:sapphire gain medium to between approximately 700 and 1000 nm.
Alternative neodymium-doped and ytterbium-doped dielectric or glass laser sources with slightly longer emission wavelengths exceeding 1 µm have also proved useful for nonlinear-microscopy applications. Indeed, both the Ti:sapphire and the 1-µm-wavelength systems have been successfully used for nonlinear microscopy, providing three-dimensional reconstructions of an imaged volume with submicron spatial resolution and better specimen preservation than in linear-microscopy methods.
Numerous recent reports, however, suggest that many nonlinear-microscopy applications can benefit from using longer-wavelength-emitting excitation sources.1-5 Femtosecond-pulse chromium-doped forsterite (Cr:forsterite) laser sources emitting around 1260 nm, for example, have found applications in deep-tissue imaging via THG and three-photon-excitation laser-scanning fluorescence microscopy (3PLSM) in a range of samples. 6, 7, 8 Unfortunately, the relatively narrow gain bandwidth of Cr:forsterite restricts the tuning range of the source to a few tens of nanometers. A tunable source delivering wavelengths longer than 1200 nm would potentially enable more efficient multiphoton excitation, and would also offer the possibility of improved SHG and THG in a wider range of samples without compromising the advantages of the Cr:forsterite source.
Wavelength-tunable alternative
Nonlinear optical-frequency conversion offers a potential solution to this technology limitation. Synchronously pumped optical-parametric-oscillator (SPOPO) sources can be engineered to provide subpicosecond radiation with moderate average power that is widely tunable at longer wavelengths. To this end, our research team at the Centre for Biophotonics developed a singly resonant SPOPO that provides a wavelength-tunable alternative to the Cr:forsterite source for nonlinear microscopy of a range of specimens and that offers the possibility for yet longer-wavelength excitation. This flexible source incorporates a periodically poled lithium niobate (PPLN) crystal within an external resonator that is synchronously pumped by a commercial femtosecond-pulse neodymium-doped yttrium lithium fluoride (Nd:YLF) laser source to provide femtosecond-pulsed continuously tunable radiation from 1.45 to 1.63 µm. Both THG and 3PLSM of samples have been performed using the long-wavelength SPOPO source.
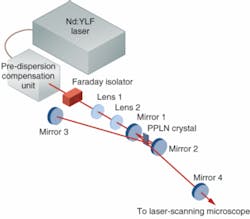
A commercial femtosecond-pulse Nd:YLF laser from Coherent (Santa Clara, CA) was used as the pump source for the SPOPO (see Fig. 1). The system comprised the oscillator and a pre-dispersion-compensation unit. While this particular laser is no longer commercially available, alternatives capable of delivering femtosecond pulses at similar wavelengths—that is, those based on an ytterbium-doped or other neodymium-based gain medium—can be used as a simple replacement. The laser delivered approximately 0.6 W of average power at a wavelength of 1047 nm (6 nm spectral full width at half maximum), with a pulse duration of approximately 406 fs and and a repetition rate of 120 MHz. This radiation was propagated through a Faraday isolator to minimize etalon effects that otherwise disrupted the modelocking process. The output from the pump laser was mode-matched to the SPOPO resonator using two antireflection-coated spherical lenses.
Tuning by crystal repositioning
The nonlinear crystal used in the SPOPO was a 3-mm-long multigrating PPLN crystal. Periodically poled lithium niobate was chosen because of its large d33 (second-harmonic) coefficient and substantial OPO bandwidth, as well as the quality and availability of crystals. The PPLN crystal was held in a homemade oven at 130°C to limit photorefractive damage to the crystal and to avoid oxygen migration into the crystal. The grating period varied from 28.5 to 29.9 µm in 0.2 µm increments. Locating the oven on a translation stage allowed the period interacting with the light and hence the wavelength to be varied by translating the crystal relative to the pump source. This was designed to provide a signal-wavelength output tunable from 1.46 to 1.60 µm, with the corresponding idler wavelength tunable from 2.8 to 3.7 µm with the PPLN held at a fixed temperature. Mechanical repositioning of the crystal is faster than temperature tuning of the crystal and also reduces the probability of thermal damage to the crystal.
Only the signal wavelength was intended for use in this study, while the idler wavelength was rejected. The mirrors in the external resonator were custom-specified to be highly transmitting at the pump wavelength of 1047 nm and highly reflecting at the expected signal wavelength range of 1.46 to 1.6 µm. The external linear resonator comprised two zero-lens mirrors with a 100 mm radius of curvature adjacent to the PPLN crystal (M1 and M2 in Fig. 1) and two plane mirrors (M3 and M4). One of the plane mirrors (M4) served as the output coupler by providing 2% transmission across the expected signal-wavelength range.
Following characterization, the output from the OPO was coupled into a laser-scanning microscope (LSM). The scan head of the LSM, from Bio-Rad (Hemel Hempstead, England), was coupled to an inverted microscope. The scan head contained a pair of scanning galvanometer mirrors to enable the line scan, and sensitive photomultiplier tubes to detect the resultant fluorescence or backscattered harmonically generated radiation from the sample under investigation. The inverted microscope was used in conjunction with a 40× magnification/1.3 numerical aperture oil objective lens; the fluorescence signal or harmonically generated radiation was collected using an “epi” configuration, in which the same lens that provides the excitation light also collects the fluorescence or harmonically generated radiation. Epi-detection for THG has already been successfully performed by Debarre and his colleagues.6Ample light
The peak intensity of the SPOPO source was ample for both 3PLSM and THG microscopy across this wide wavelength range. Furthermore, during the imaging of fixed and live samples, the samples did not appear to be harmed as a consequence of imaging. This agrees with the work of the Debarre team and Sun et al., in which longer-wavelength sources were reported to preserve cell viability.6, 7 Furthermore, the wide tuning range of the SPOPO means that the excitation wavelength can be matched to the absorption of the fluorophore or to perform more-efficient THG.
At the shorter wavelengths available from the SPOPO, three-photon and THG signals were both weaker at the same average power than when tuned to wavelengths longer than 1.4 µm. From this, we conclude that while the Cr:forsterite laser can be used for 3PLSM and THG imaging, the longer wavelengths from the SPOPO are better suited to these applications.
The ability to tune the excitation wavelength enables a reduction in average power delivered to the sample while maintaining very high peak intensity through the ultrashort-pulsed nature of the source. Although this peak intensity is extremely high, it is unlikely to be sufficient for four-photon processes. However, the wavelength range of the SPOPO is suited to simple pulse-compression methods.9, 10 Our team now plans to investigate the possible application of the SPOPO for yet higher-order processes.
REFERENCES
1. Y. Barad et al., Appl. Phys. Lett. 70, 922 (1997).
2. M. Muller et al., J. Microsc 191, 266 (1998).
3. J.A. Squier et al., Opt. Express 3, 315 (1998).
4. A.C. Millard et al., Appl. Opt. 38, 7393 (1999).
5. L. Canioni et al., Opt. Lett. 26, 515 (2001).
6. D. Debarre et al., Nature Methods 3, 47 (2006).
7. C.K. Sun et al., J. Struct. Biol. 147, 19 (2004).
8. H. Matsuda et al., J. Phys. Chem. Lett. B110, 1091 (2006).
9. O.E. Martinez, IEEE J. Quant. Electron. 23, 59 (1987).
10. G. McConnell and E. Riis, J. Biomed. Opt. 9, 922 (2004).
GAIL McCONNELL holds a Research Councils U.K. Academic Fellowship at the Centre for Biophotonics, SIPBS, University of Strathclyde, 27 Taylor Street, Glasgow, Scotland, G4 0NR; e-mail: [email protected]; www.strath.ac.uk/sipbs.