CELLULAR IMAGING: Novel nonlinear contrast improves deep-tissue microscopy
WARREN S. WARREN, MARTIN C. FISCHER, AND TONG YE
For centuries, the optical microscope has been the premier workhorse tool for quantitative understanding of cellular and tissue structure at the submicron level. But conventional microscopy has some important limitations; in particular, the human body is relatively opaque in the visible.
Although penetration depths in the red and near-infrared (650-1300 nm) range up to many centimeters, near-IR imaging suffers two fundamental problems: scattering and lack of contrast. Photons are typically scattered 30 to 50 times before being absorbed, leading to highly blurred images. Several methods can be used to alleviate this scattering problem (for example, confocal microscopy, ultrasound-mediated optical tomography, and optical-coherence tomography), but the subcellular resolution needed for diagnostic purposes remains elusive except very near the surface. In addition, contrast arises from only a few biologically interesting molecules.
Nonlinear signatures
Over the last decade, the use of nonlinear signatures has evolved as a versatile solution to the issues of spatial resolution and molecular contrast. The most developed of these signatures create light at a new wavelength, which can then be separated from the exciting laser. For example, molecules can absorb two photons in the near-IR to access states at energies that would require UV photons for direct absorption. The excitation can be easily detected if the molecules fluoresce and if the two-photon fluorescence (TPF) can get back out of the sample. High spatial resolution is made possible by the quadratic dependence on laser intensity—the fluorescence signal is generated almost entirely in the laser’s focal volume, which can be as small as a femtoliter. While TPF can be induced by continuous-wave lasers, the vast majority of studies use short (less than 100 fs) laser pulses to achieve high peak power with relatively low tissue damage from average-power dissipation.
Naturally occurring fluorescence originates from molecules such as NADH, flavins, elastin, collagen, and porphyrins; in many applications, however, this contrast is insufficient, and instead is enhanced by chromophores that are injected (dyes) or expressed (green fluorescent protein and its variants). Second-harmonic generation (for example, direct generation of 400 nm light from 800 nm excitation) only takes place in anisotropic media; thus, such images primarily highlight collagen, like connective structures in the dermis. Third-harmonic generation has also been reported, but very high powers are needed. Coherent anti-Stokes Raman microscopy (CARS) excites a vibrational transition with two pulses, and then uses this coherence to shift the color of a third pulse. Unfortunately, CARS gives a relatively weak signature, and most of the microscopy work has focused on the carbon-hydrogen stretching frequency (which is ubiquitous in biomolecules).
Novel nonlinear contrast
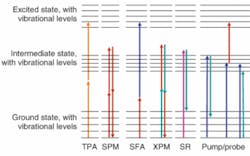
Recently, we have been focusing on other nonlinear signatures that are already well known in physics and engineering. The most important single-wavelength effects for our purposes here are two-photon absorption (TPA) and self-phase modulation (SPM; see Fig. 1). Melanin (which is actually a range of biological pigments found in a variety of locations including the hair, skin, eyes, brain, and inner ear) is a good example of a TPA target; it is not a suitable target for imaging methods based on fluorescence because of its extremely low quantum yield (less than 10-3), strong fluorescence quenching and reabsorption, and opacity in the visible region.
Self-phase modulation arises because the index of refraction can have a contribution proportional to the instantaneous pulse intensity, most commonly due to Kerr nonlinearities. The SPM coefficients of common neat solvents vary over a factor of more than 100; the phenomenon is highly sensitive to localized structure, and appears to change markedly when neurons fire.1 Dual-wavelength experiments (with or without a real intermediate state), including sum-frequency absorption and cross-phase modulation, add additional control parameters (wavelengths, interpulse delay, polarization dependence) that can distinguish contributions from different biomarkers (for example, eumelanin from pheomelanin).
The challenge with all these signatures is that, in the power limits we care to use on tissue (on the order of 1 mW from a modelocked laser), they do not ordinarily generate light at a different frequency from the laser itself, making detection challenging. For example, TPA can in principle be detected by the attenuation of the laser beam, but at the power levels we use, perhaps 10-6 of the photons are taken up by this mechanism; the rest of the photons are lost to linear absorption and scattering, which also attenuate the beam. To make things worse, laser pulse amplitudes have jitter, and subjects move. Not surprisingly, then, these novel nonlinear signatures have been largely ignored.
Pulse and pulse-train shaping
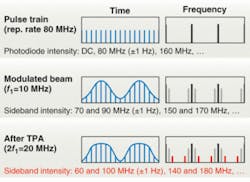
The trick that makes these signatures observable is simple in principle: force them to create light at a different frequency. We do this by shaping the laser pulse train (see Fig. 2), the individual laser pulses, or both.2, 3 For example, a laser modelocked at 80 MHz generates a precise frequency comb with detectable intensity components at n × 80 MHz, where n is limited in practice by the bandwidth of the photodetector. Clean amplitude modulation (for example, at a frequency f1 = 10 MHz) will impose one upper and one lower sideband offset by f1 from each of these lines.
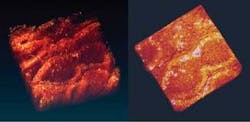
Two-photon absorption is more effective on the intense pulses than on the weak ones, and thus it distorts the amplitude modulation, creating extra sidebands at ±2f1. Only a nonlinear process can create this sideband, so the signal is no longer swamped by competing effects. In one example of such a measurement, melanin in lesions induced on human skin tissue is imaged (see Fig. 3). Two modelocked lasers (modulated at f1 and f2 respectively) create sidebands at f1 ± f2. The modulation frequencies in general are chosen to minimize interference from physiological noise (which extends into the kilohertz regime) and laser noise.
Self-phase modulation cannot be detected in this manner because it does not actually attenuate the laser pulse. However, it does alter the pulse spectrum. In fact, at high powers, it is the dominant mechanism in white-light-continuum generation, which is used to seed the first stage in many optical parametric amplifiers. At the powers we use, reshaping effects are very subtle, unless you start with a shaped pulse.
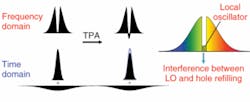
Consider a laser pulse with a “hole” in the center of its spectrum (see Fig. 4). The Fourier transform shows that such a pulse has zero area in the time domain-that is to say, it consists of a short and intense pulse on top of a weak, negative, longer pulse. Because TPA is proportional to the square of the intensity, the intense pulse is attenuated more than is the weak pulse. The consequence of this is that after TPA the area is no longer zero, and the hole tends to refill (180° out of phase with the outside of the pulse). Hence, frequency-resolved detection can find the weak TPA signal on top of a very low background. Self-phase modulation will also refill the hole, but in that case a similar figure would show the refilling is 90° out of phase.
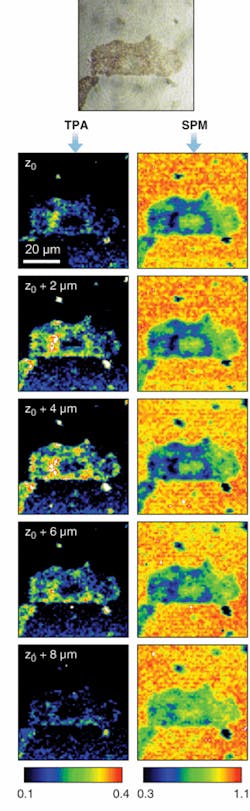
Of course, detecting the phase of an optical field is very difficult; however, if instead of a perfect hole we leave a weak field and vary the phase of that field, we can observe the refilling by interference. Acousto-optic pulse-shaping technology can produce very complex waveforms, and more important for our purposes, can alter the pulse shape fast enough to beat physiological noise.4 In an example, SPM and TPA images, in this case of a mounted B16 melanoma cell, are simultaneously acquired (see Fig. 5).
In all these cases, the laser pulses and the acquired signal (transmitted or backscattered light) are in the wavelength regions for which tissue is relatively transparent, although scattering still limits the depth at which high peak powers can be achieved. In practice, this means that microscopic resolution to depths of about 1 mm should be achievable in most tissue—one to two orders of magnitude farther than conventional microscopy, and with far more physiological contrast. Important applications are likely to include melanoma diagnosis, evaluation of surgical margins, and characterization of the region around an endoscope.
REFERENCES
1. M.C. Fischer et al., Proc. SPIE 6442, 64421J, (2007).
2. P. Tian and W.S. Warren, Optics Letters 27, 1634 (2002).
3. M.C. Fischer et al., Optics Letters 30 (12) 1551 (2005).
4. J.X. Tull et al, Advances in Magnetic and Optical Resonance 20, 1 (1997); A.M. Weiner, Rev. Sci. Instrum 71, 1929 (2001).
Warren S. Warren is the James B. Duke professor of chemistry, radiology, and biomedical engineering; Martin C. Fischer and Tong Ye are assistant research professors in chemistry at Duke University, Durham, NC 27708; e-mail: [email protected].