Self-interference method yields sensitive spectral microscopy
Researchers at Boston University have successfully demonstrated a microscopy technique that may enable subcellular optical imaging with resolution on the order of a few nanometers.
"There are a lot of very nice techniques to get below the standard diffraction limit," said Bennett Goldberg, who co-authored a paper presented at Photonics West (San Jose, CA) in January along with co-team leaders Anna Swan and Selim Unlü. "And I would say the absolute best state-of-the art resolution that people get with fluorescence microscopy is typically about 100 nm or so."
The Boston researchers, however, were seeking ways to go significantly beyond this apparent resolution limit for real-time imaging of biological phenomena such as protein location and transfer in live cellular systems.
"You could think of all sorts of trafficking issues in cellular cycles that are incredibly important," Goldberg said. "You can also think of things like transcription itself, which depends on protein collocation. None of these has ever been imaged on any sort of real-time basis."
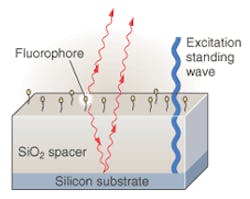
About a year ago, their efforts paid off in the form of a new technique that they call "spectral self-interference." It involves placing a mirror within relatively close proximity (5 to 10 μm) to the fluorophore, yielding an interferogram between direct and reflected fluorophore emissions (see Fig. 1). Previous researchers had noted dramatic constructive and destructive interference effects when the distance between fluorophore and mirror were on the order of one wavelength. But by increasing the distance to the order of 10 wavelengths, the Boston team observed a very sensitive, spectral position indicator.
"The smooth spectrum that you had expected to see from a fluorophore is now a spectrum with sinusoidal oscillation in it," Goldberg said. "And that sinusoidally oscillating spectrum tells you very precisely the distance between the fluorophore and the mirror."
In initial experiments, the researchers placed fluorophores on a silicon dioxide surface elevated five or 10 μm above a silicon surface, which tended to behave like a mirror because of the relatively high reflectivity at the interface between the two materials. Then they bound a monolayer of fluorescein to the silicon dioxide surface adjacent to a protein (streptavidin) monolayer on the same surface, also beneath a monolayer of fluorescein. The resulting spectral self-interference image clearly distinguished the approximately 5-nm height difference between the two adjacent fluorescein layers (see Fig. 2)."Basically what you are doing is encoding the height information in the spectrum," Goldberg said.
Of course in an actual biological context, one would want to look at several different fluorophores with arbitrary spatial distributions. The researchers intend to deal with this natural complexity by combining the new self-interference technique with a relatively well-known technique called standing wave interference that enables differential spectral weighting of different fluorophores at different heights.
"We've actually worked out ways of taking a minimum number of positions of the standing wave and then deconvoluting the output spectrum to determine the vertical position of the fluorophores, even with an arbitrary distribution in the vertical plane," Goldberg said. He also expects the method to extend easily into three-dimensional observations.
"Ultimately we believe this will allow biologists to do some very interesting subcellular microscopy," he said.
About the Author
Hassaun A. Jones-Bey
Senior Editor and Freelance Writer
Hassaun A. Jones-Bey was a senior editor and then freelance writer for Laser Focus World.