PLASMONIC OPTICAL STRUCTURES: Tilted FBGs excite optical-fiber plasmons
Jacques Albert and Yanina Shevchenko
Plasmon waves come from collective excitations of electrons occurring at the boundary between a metal and a dielectric. The strong sensitivity of the plasmon-propagation constant to the permittivity of a nanoscale layer of material on a metal surface has led to the development of an exquisitely sensitive technique called surface-plasmon resonance (SPR) to detect chemical and biochemical changes occurring between molecules. Laboratory instruments based on this technique have been developed for important applications in drug discovery, toxicology, and biochemical research.
A miniature variant of the standard SPR configuration has been developed that is based on a metal-coated ordinary optical fiber in which a refractive-index grating has been written using intense UV light. This optical-fiber SPR device is extremely easy (and relatively inexpensive) to fabricate and to interrogate. The use of a standard communications fiber allows the physical separation of the transducing function (converting a biochemical reaction into an optical property change) from the interrogation mechanism even over large distances. We hope this new platform will enable the development of a new class of biochemical sensors that are capable of remote monitoring, that can be distributed or inserted into small spaces, and that can be developed into a very low-cost alternative to current instrumentation.
Sensing with plasmon excitations
Biochemical reactions are measured with plasmon waves by monitoring their effective index (the ratio of the propagation constant of the wave in the metal to the propagation constant of light at the same frequency in vacuum). In practice, this is done by launching light beams from a high-refractive-index medium toward an interface coated with a thin metal layer at angles such that the light is totally reflected. When the component of the propagation constant of the light along the interface matches that of a plasmon excitation of the other side of the metal layer, some of the light couples to the lossy plasmon and the reflection diminishes. Propagation constants are scanned either by changing the wavelength and keeping the incidence angle constant or by using monochromatic light and scanning the angle.
In all cases the light must be polarized parallel to the plane of incidence to satisfy the requirement that the plasmon wave be polarized normally to the interface. Biochemical reactions occurring at the surface of the metal change the effective index of the plasmon and can be detected as a shift of the resonance condition. Recent improvements to this technique have used interferometers and gratings to increase the sensitivity and accuracy of the measurements.1
Over the last 15 years or so, various implementations of SPR sensors on optical fibers have appeared. The most straightforward consists of using a multimode fiber with a section of cladding removed or thinned sufficiently for core-guided light to interact with a metal coating on the fiber. The presence of a plasmon resonance then shows up as a dip in the transmission spectrum. This can be improved by using single-mode fibers that are D-shaped or side-polished to expose the evanescent field of the core-guided light to a metal coating deposited on the flat side. However, the etching or polishing must be done with high accuracy to control the amount of evanescent-field overlap; in addition, the process weakens the fiber.
FBGs for resonant coupling
Our approach for fiber SPR uses a grating-assisted concept, as well as polarization control, to generate optical plasmons that are unambiguous and easy to measure in unmodified cylindrical single-mode fibers. The great simplicity of our concept lies in the fact that, instead of patterning the fiber surface or the metal layer, we use fiber-Bragg gratings (FBGs) photoinscribed in the core of the fiber to achieve resonant coupling to the plasmons. The only modification to a standard fiber grating is a small tilt in the orientation of the grating planes relative to the axis of the fiber. This tilt has two functions: enhancing the coupling of the light from the core to the cladding modes and breaking the cylindrical symmetry of the coupling process relative to the state of polarization of the guided light. The idea is derived from a tilted-FBG (TFBG) refractometer invented by Laffont and Ferdinand in 2001.2 The TFBG coupling mechanism resembles the standard prism-based SPR sensor configuration (see Fig. 1).
The fiber used in all our work so far is Corning SMF28 single-mode telecommunications fiber. The fiber is hydrogen-loaded to enhance its photosensitivity; we use an excimer laser operating at a wavelength of either 193 or 248 nm to write Bragg gratings in the fibers by the phase-mask technique.
Typical experimental transmission spectra of a TFBG with a 10° tilt show dips in the spectra. These dips correspond to light coupled out of the core mode into one of the modes guided by the cladding (or back-reflected in the core for the rightmost resonance). The effective indices of the cladding modes excited by this grating range from 1.2 to 1.44. When the exterior refractive index increases above the effective index of a given set of cladding modes, the modes becomes radiative and their resonance disappears from the spectrum. This is the principle of the TFBG refractometer. A useful feature of the TFBG is that the core mode back-reflection resonance (the resonance with the longest wavelength in the experimental spectra) is totally insensitive to the external medium and can be used as a power and wavelength reference in addition to providing an in situ thermometer (a well-known application of standard FBGs).
To transform the TFBG into an SPR sensor, all that is required is a thin metal coating on the surface of the cladding. So far, we have used a crude approach consisting of sputtering gold in two steps separated by a 180° fiber rotation around its axis, for an average thickness of about 50 nm. The sensor can be operated using unpolarized light.3 However, a polarization controller inserted anywhere between the light source and the sensor improves the SPR clarity significantly (as shown in the experimental spectra) for a gold-coated TFBG in water.
Polarization effects
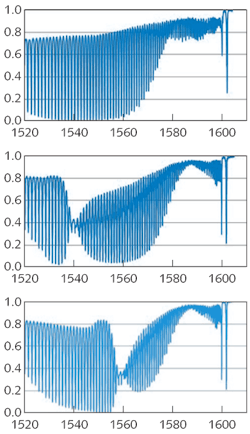
By scanning the polarization angle of the linearly polarized light guided by the core, we clearly find two orthogonal orientations for which a plasmon resonance is either absent (Fig. 2, top) or maximized (Fig. 2, center). The SPR resonance of these spectra corresponds to the most attenuated cladding-mode resonances—that is, those that are phase-matched to a plasmon wave of the metal coating. The polarization can be adjusted at any time to ensure maximum SPR visibility following displacements of the sensor head or of the interrogating fiber. Furthermore, the SPR shifts when the external medium is changed, as for the same grating immersed in ethanol (Fig. 2, bottom). In both media, the position of the measured SPR corresponds to the expected resonance wavelengths calculated for this structure using a commercially available finite-difference cylindrical-mode solver (Fimmwave by Photon Design; Oxford, England).A full characterization of the SPR sensor can be carried out by measuring the SPR wavelength while immersing the grating in liquids with various refractive indices: these sensors have a measurement range that is bounded by the refractive index of the glass used (1.44) and the effective index of the highest order cladding modes that can be excited by the TFBG (about 1.2). Therefore, measurements in gases are not possible at the moment unless a reactive coating is used to transform gas concentrations into measurable refractive-index changes in the layer adjacent to the metal.
The sensitivities observed so far range from 450 to 1500 nm/(refractive index unit) and the minimum detectable level that we measured corresponds to a refractive index change of 5 × 10-5. This is two orders of magnitude worse than the best commercially available bulk SPR sensors, but at a cost (and size) that is two orders of magnitude smaller. It is worth noting that we have not yet even attempted to use the spectral shifts of the individual resonances near the SPR to fine-tune our measurements and improve the accuracy: there is a large quantity of data available from the positions and amplitudes of the fine comb of cladding-mode resonances in the spectrum.4
Measuring protein concentrations
Finally, since the main application of SPR sensing technology is biochemistry, we have been carrying out preliminary tests of the ability of our simple device to measure the binding of nucleic acids and proteins on the gold-coated surface of the TFBG.5 To achieve molecular selectivity, the gold surface is first coated with a layer of thiolated aptamers (single-strand DNA molecules with a binding group for gold at one end and a receptor molecule for a protein called thrombin at the other end).
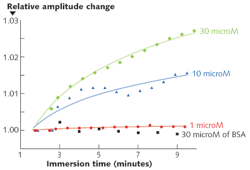
The sensor's responses to the attachment of various concentrations of thrombin in solution were measured (see Fig. 3), and was compared to its response to a control solution containing the "wrong" protein (bovine serum albumin, or BSA). In this case the SPR shift was inferred from the amplitude shift of an adjacent cladding-mode resonance. We are still in the process of characterizing the measured responses and optimizing the protocols for such experiments, but the results are a clear demonstration of the detection capabilities of our sensors for events occurring in relatively dilute nanometer-thick biomolecule monolayers that partially cover the surface of the fibers.The sensor can be fabricated by anyone who has access to an FBG-writing facility and to some means of depositing metals into thin films; the measurements require only standard photonic-component instrumentation. As a result of this simplicity, we hope that many people will try fiber-plasmonic biosensing and that low-cost tools will be developed for applications in which large and expensive laboratory instruments cannot be used. It is also intriguing to think of applying this platform to other novel biophotonic technologies, such as the excitation and collection of fluorescence or enhanced Raman scattering from nanoparticles dispersed on the surface of a fiber. We also believe that more-sophisticated data processing will eventually lead us to take advantage of the large amount of information that is present in the TFBG spectrum.
REFERENCES
- M. Piliarik and J.Homola, Opt. Express 17, p. 16505 (2009).
- G. Laffont and P. Ferdinand, Meas. Sci. Technol. 12, p. 765 (2001).
- Y. Y. Shevchenko and J. Albert, Opt. Lett. 32, p. 211 (2007).
- B. Spackova et al., Sensors and Actuators B 139, p. 199 (2009).
- Y. Y. Shevchenko et al., Conf. Lasers and Electro-Optics, paper CMJ4 (2008).
Jacques Albert holds the Canada Research Chair in Advanced Photonic Components and Y. Shevchenko is pursuing a Ph.D. degree in the Department of Electronics, Carleton University, Ottawa, ON, Canada; e-mail: [email protected].