In 1983, with oil prices sky high, solar energy seemed the wave of the future, and forecasters predicted annual rates of growth of 50% in the solar-cell market. Things didn’t turn out that way—solar energy remained far more expensive than other forms of energy, and sales grew at a rate of 12% instead. Today, solar energy remains limited to a number of important, if small, niche markets: space power, small portable devices such as calculators, larger devices such as vaccine refrigerators, and power for remote areas.
Over the past few years, however, technological advances in thin-film cell design have again raised hopes for a major decrease in cost and a large expansion of the market. Solar cells may still be too expensive to compete directly with the main energy sources for utility grids, but their use in other applications may significantly increase.How solar cells work
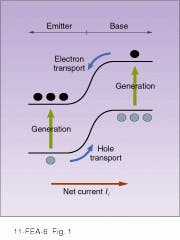
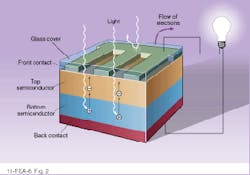
Like other photodetectors, solar cells convert light energy into electrical energy, using the same basic principle as p-n diodes. Photodiodes, including solar cells, consist of a p-n junction of two semiconductor types, which allows current to flow in only one direction (see Fig. 1). The impurities or dopants create a positive charge on one side and a negative charge on the other, generating a potential across the junction. If a photodiode is operated without a bias potential—that is, without an additional external voltage--it becomes a photovoltaic device. When such a cell is exposed to light, photons bounce electrons into the conduction band, creating electron-hole pairs. The intrinsic potential separates the charges, and they accumulate on opposite sides of the cell. This in turn generates a voltage from which a current can be derived. In solar-cell modules, electrodes laid over the top of the cells collect the current and feed it out to the cell edge (see Fig. 2).Solar energy is an extremely dilute source of power with a maximum of about 1 kW/m2. Averaged over the earth`s surface and over the whole year, day and night, solar radiation amounts to only about 340 W/m2. So, even at 100% efficiency a solar power station with 1000-MW capacity would need 3 km2 of solar cells.
Unfortunately there are a number of inherent losses in solar cells that lower their efficiency in converting light to electric power. First, the solar power is spread over a considerable range of wavelengths, and only those photons with energies above the bandgap—the energy gap between the valence and conduction bands—will be absorbed in any given semiconductor. Below-bandgap photons, with typically 20% of total light power, are lost. Second, even those photons that have a short enough wavelength are not fully utilized because once an electron-hole pair is created, each carrying more than the bandgap energy, the charges lose energy to decay down to the bottom of the conduction band. This lost energy, another 30% of the incident total, goes into heat and is also unavailable for electric power. In addition, some light may be reflected from the top surface, and other light may not be absorbed, but passed through the semiconductor material. The recombination of electron-hole pairs causes further losses.
The net result is that, in general, the best laboratory cells achieve at most some 25% efficiency, while production cells barely exceed 10%. This modest efficiency, combined with the dilution of solar energy, has made solar energy considerably more expensive than fossil and nuclear power. System costs, including the cells themselves, support and protective structures, and controls, remain in the area of $8/W, about a factor of 10 above conventional large-scale power-grid sources.
From space to earth
Despite their expense, solar cells have found wide use in applications where other power sources are impractical. The oldest and best known of these is the supply of electrical power in space. Except for a relatively small number powered by nuclear sources, nearly all spacecraft today are powered by solar cells, and the need for such a power supply was the main impetus for government funding of solar-cell development in the 1950s and 1960s. For this use, the safety and relative light weight of solar cells overshadow their cost, which tends to be a small fraction of total spacecraft cost.
On earth, solar cells are used as an alternative to batteries in portable devices such as calculators or in remote sites where the expense of delivering electricity far exceeds the expense of producing it (see photo at top of this page). Remote systems used for small-scale industry, water pumping, and village lighting (primarily in Third World countries) are now the main market for photovoltaics.
Solar cells are also used for powering remote telecommunication repeater stations, and there is a large potential for market growth in providing power for portable vaccine refrigerators, a vital component of the United Nations World Health Organization`s vaccination programs in poor nations. Due to their still-high costs, cells for residential power and for utility supply remain very small sectors of the market.
From crystals to thin films
There are two basic types of solar cells today: older crystalline-silicon type and newer thin-film types, of which there are many. Crystalline silicon is the basic material of the semiconductor industry and is a relatively insignificant cost component of integrated circuits. But the very large area of solar cells makes silicon costly in both money and energy. Silicon wafers typically cost around $1500/m2, which immediately places solar cells based on this material in the range of $10-$20/W.
In addition, crystalline silicon is energy-intensive to produce. A typical Earth-based solar cell must operate continuously for about two months merely to replace the energy consumed in its production. However, crystalline cells make up for these disadvantages by their higher efficiency, as much as 18%, and their reliability. In addition, gallium arsenide crystalline solar cells, used for some space and defense applications, have high resistance to ionizing radiation.
To overcome the expense of crystalline solar cells, great effort is being focused on the development of thin-film cells. Crystalline silicon cannot be used as a thin film, because it is an indirect absorber--absorption requires the interaction of a photon with a phonon, a unit of vibrational energy—so relatively thick layers are needed. Amorphous silicon, however, with many different bond lengths, is a direct absorber and is highly opaque, so it can be used as a thin film. But there is a problem. Amorphous silicon is not a true semiconductor because the energy gap between the valence and conductance bands is actually filled with intermediate states.
The solution to this problem was found in 1969, when hydrogen was added to reduce the number of intermediate states and create a semiconductor. The use of amorphous silicon thin films in solar cells developed slowly, because light creates defects in amorphous silicon, which reduces efficiency as exposure to light continues. Stabilized amorphous silicon, after long light exposure, had efficiency of around 6%, well below crystalline silicon.
A second approach uses polycrystalline silicon, consisting of many small crystals, which is far less expensive to produce than single-crystal wafers. While this form also has low absorbance, in a thin film with a rough backing multiple internal reflections can increase the light path length twentyfold, leading to good absorption in films only tens of microns thick. (Monocrystalline silicon requires flat substrates for regular structure, so this technique can`t be used.) Such polycrystalline films have achieved 12% efficiency in commercial cells.
Two other materials are used in thin films. Both copper indium diselenide (CIS) and cadmium telluride (CdTe) can be used to form heterojunctions with cadmium sulfide (CdS). Both of these materials have efficiencies in the 9%-12% range (see Fig. 3). Recent developments at the National Renewable Energy Laboratory (NREL; Golden, CO) have produced CIGS thin-film cells with efficiencies greater than 17% (see Laser Focus World, July 1996, p. 11)Besides the use of thin films, there is another way to decrease solar electric costs: concentrating the light first. Concentrating systems are generally only practical for reasonably large-scale applications, especially production of utility grids, and have been the focus of research funded by organizations such as the Electric Power Research Institute (EPRI; Palo Alto, CA). Concentrating systems using Fresnel lenses, parabolic mirrors, or heliostats (col lections of flat mirrors all oriented toward a single area) can increase light intensity five-hundredfold. Relatively cheaper lenses and mirrors, rather than the more-expensive solar cells, serve as the large-area collectors and are the main determinants of system cost.
EPRI studies have estimated that concentrator systems can eventually be produced for as little as $2/W. This is still twice the capital cost of other sources and would probably lead to the production of higher-cost electricity, an indication of how difficult achieving inexpensive solar power really is.
Advances in thin-film technology
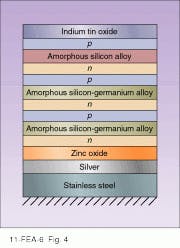
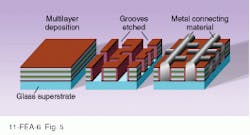
In 1994, the prospects for amorphous silicon cells brightened considerably with the development of two different approaches to increasing efficiency. United Solar Systems Corp. (Troy, MI), with support from the NREL, developed a device that achieves stabilized efficiency of greater than 10% by making the initial collection of light more effective. The core of the device is three different p-n junction layers—the top is amorphous silicon and the other two are amorphous silicon-germanium alloys, each with different doping levels of germanium (see Fig. 4). The result is that each layer has a different bandgap and therefore absorbs different parts of the solar spectrum, thus minimizing excess energy losses. The top layer slices off the highest energy photons with a large bandgap, and the next two layers take progressively lower-energy photons, in each case minimizing the amount of energy lost to heat. A transparent indium tin oxide layer on top doubles as an electrode and antireflection coating. The whole sandwich is laid down on a thin stainless-steel substrate, making the solar cells flexible.A second approach developed by University of New South Wales (Australia) researchers Martin Green and Alistair Sproul also uses multiple layers, but, in this case, the layers are of ordinary n- and p-doped silicon. The trick is that each layer has its own separate electrodes. The electrodes, rather than running across the top of the thin film, are buried within in it, running top to bottom (see Fig. 5). Alternating sets of electrodes contact n and p layers. In this way, efficiencies as high as 25% with costs as low as $10/m2 are claimed. But much work is needed before such laboratory devices can be commercialized.
Solar energy cells that can compete directly with fossil and nuclear power appear to remain beyond the horizon. However, the new generation of solar cells is likely to make photovoltaics a rapidly growing market in the coming years.
About the Author
Eric J. Lerner
Contributing Editor, Laser Focus World
Eric J. Lerner is a contributing editor for Laser Focus World.