Fiber Lasers: Raman-scattering-based gas analysis in metallurgy, medical, and food industries
The advancement of gas-analysis techniques has been a focus of research efforts for decades.1, 2 The two most compelling and selective methods traditionally used in this field are chromatography and mass spectrometry. While these methods work very well, they have a number of drawbacks: bulk and complexity of equipment, considerable time needed for analysis, and the need to place a sample inside the equipment chamber. These drawbacks impede the use of such equipment for online and remote monitoring.
Laser-based gas analysis techniques can optimally answer these challenges. Laser methods yield prompt results and enable measurements both locally and at considerable distances using a reflected signal. However, laser-based methods can be inferior in both selectivity and sensitivity, and can fail to discriminate between different materials with similar characteristics.
Laser methods can be divided into three main branches:
• Laser absorption
• Recombination (Raman) method
• Resonant fluorescence
These methods essentially differ in the intensity of the processes that yield valid signals. The strongest is the absorption method. For example, the absorption cross section of sulphur dioxide (SO2) is 10–20 cm2, for Raman scattering it is 10–30 cm2, and fluorescence techniques rank between these two values. These are significant differences—as a result, Raman spectroscopy is much less suited for gas analysis when compared to absorption techniques.
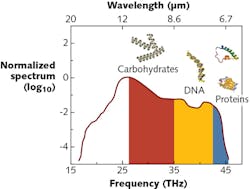
The spectral range is an important design factor: ultraviolet (UV) and visible-light photons have higher energy than electron transitions and thus can sense materials as a result of these transitions. Conversely, a spectral range in the mid-infrared (mid-IR) yields photons with lower energy—therefore, their interaction with matter will affect molecular vibrations (spectral range from 3 to 14 μm) rather than electron transitions (see Fig. 1). For even longer wavelengths, transitions are too weak to practically consider.
Comparing absorption-spectroscopy methods
We will consider several examples of gas analysis via absorption-spectroscopy methods. One example is a device to detect sulfur hexafluoride (SF6) leaks from electrical installations, the LaserGasTest, made by Hypertech Laser Systems (Lübeck, Germany).9 This device uses the photoacoustic effect for operation. Principal atmospheric gases, such as nitrogen and oxygen, are absorptively inert in the mid-IR range; thus, a tube filled with pure nitrogen will not absorb laser radiation in the mid-IR. Adding a laser-absorptive gas (SF6, in this case) to the tube causes partial absorption of photons passing through the tube. Upon absorbing laser radiation, the SF6 gas heats up along the beam trajectory and expands. If a microphone is mounted on the side wall, the corresponding acoustic signal can be detected, with its strength correlating to the gas concentration. Such a device allows detection and quantitative characterization of the gas.
Following this principle, a team from the Institute of Laser Physics (Novosibirsk, Russia) and colleagues from the Institute of Atmospheric Optics of the Russian Academy of Sciences (Tomsk, Russia) have designed a device for electronegative gas (SF6) analysis with detection accuracy up to several parts per billion (ppb). This is an extremely useful invention for high-voltage applications where electronegative gases are used, as it reduces leak-detection time from one month to one hour, even providing the possibility of localizing the leaks.9
Currently, gas-analysis techniques for medicine are under active development—for example, with applications in patient respiration analysis and diagnostics. A considerable number of gases can be detected by the absorption method in the mid-IR range. However, the industry has lacked tunable lasers that can encompass wide spectral bands to scan a gas specimen over the entire available IR spectrum with smooth rather than discrete transitions between wavelengths.
One widely tunable light source that provides a solution is a parametric light generator with a nonlinear-crystal medium. This technology yields low radiation power, but it can be smoothly tuned across multiple wavelengths—its low power output appears to be sufficient for respiration air analysis. The tunable range of this light generator is from 2 to 11 μm.
Respiration analysis is not new—chromatographic gas-analysis methods, with all their pros and cons, have been used for this purpose until now. However, laser-absorption methods have a much higher processing rate, allowing for faster analysis. While laser absorption methods have not yet reached the stage of clinical development, they have been proven to produce reliable results. For now, we are facing the following challenges: increasing the power output for certain wave bands where it is currently insufficient; completion of comprehensive clinical tests; and development of application methods and approaches.
Raman-scattering spectroscopy
We have given a perspective on laser-absorption spectroscopy methods for various applications—let us now consider Raman-scattering spectroscopy. As mentioned before, because of the much smaller absorption cross-section of this technique and the consequent higher difficulty of generating a valid signal with an appropriate accuracy, the Raman technique requires much higher concentrations of gases for the analysis. As a result, the sensitivity of Raman-scattering spectroscopic methods is far from reaching even to several parts per million (ppm), not to speak of ppb sensitivity. This explains why the active development of Raman spectroscopy is occurring first of all in the analysis of liquids and solid particles, rather than gases.
As examples of Raman spectroscopy applications in the food industry, we can mention novel methods of determining pathogens in food products3 and implementation of Raman spectroscopy in ISO standards.4 This approach has also found application in medicine—for example, for noninvasive blood analysis,5 which can be useful for patients with saccharine diseases, and for novel methods of lung cancer diagnostics, which are more effective in early-stage detection.6
The metallurgy industry has not yet recognized the advantages of Raman-scattering techniques in comparison to laser-induced breakdown spectroscopy (LIBS) or laser-induced plasma spectroscopy (LIPS). However, Raman spectroscopy is used in scientific research into metals and their chemical contents, as well as in ore analysis for determination of its mineral composition. These problems are of importance for the field of geology.
Despite ongoing development of Raman spectroscopy for practical applications, its use in gas analysis still remains a specific and purely academic domain of interest. A group of German scientists has applied this method for monitoring gas composition and gas-exchange processes in a critical mineral exploration zone in Central Germany. In a published report,7 the scientists comprehensively described their experience of using Raman spectroscopy for oxygen (O2) and carbon dioxide (CO2) logging at depths of up to 70 m in oil wellbore columns.
Before concluding, we should mention fluorescence and light-scattering techniques for gas analysis. Flow cytometry is an excellent example for our purposes. From this perspective, we should mention the advances within the fiber-laser industry in this domain. For example, green-emitting fiber lasers have been compared for this use to conventional industrial laser light sources—namely, green and yellow diode-pumped solid-state lasers (DPSSs) emitting at wavelengths of 532 and 561 nm.8 In this comparison, an Optromix TDY-550-FS fiber-laser system was used for the research (see Fig. 2).
The use of DPSS lasers poses natural restrictions related to the operating wavelengths. The 532 nm lasers emit close to the relevant fluorescent band, so optical modifications must be made to the design for fluorescence and green fluorescent protein (GFP) detection. Solid-state 561 nm lasers also operate close to the phycoerythrin (PE) detection band, and in addition induce parasitic excitations of allophycocyanin (APC), requiring high crossbeam compensation to decrease the spectral overlap in the PE-tandem dye conjugate.
Practical application of these laser-based analysis methods poses a multitude of challenges in cases where rapid online analysis is required. For gas analysis, however, absorption-spectroscopy techniques are the most suitable solutions. Nevertheless, if both liquids and gases are to be analyzed, then both fluorescent and Raman-based techniques should be used.
REFERENCES
1. J. J. Thomson, Rays of Positive Electricity and Their Application to Chemical Analysis, London: Longman’s Green and Company (1913).
2. A. Walsh, Spectrochim. Acta, 7, 108–117 (1955).
3. T. Yaseen et al., Trends Food Sci. Technol., 62, 177–189 (Apr. 2017).
4. E. Witkowska et al., Anal. Bioanal. Chem., 409, 6, 1555–1567 (Feb. 2017).
5. R. Pandey et al., Acc. Chem. Res., 50, 2, 264–272 (Jan. 10, 2017).
6. H. Zeng, J. Biophoton., 10, 1, 98-110 (Jan. 8. 2016).
7. A. Sieburg et al., Analyst, 18, 142, 3360–3369 (Aug. 30, 2017).
8. W. G. Telford et al., Cytometry Part A, 75, 1031–1039 (2009).
9. V. A. Vasilyev, A. I. Karapuzikov, A. A. Karapuzikov, and I. V. Sherstov, "Laser optical-acoustic gas analyzer," patent for utility model No. 90905, Russia (RF)/applicant and patent holder: Special Technologies LLC.