Phosphor-converted LEDs take on industrial applications
Near-UV excitation offers high efficiency and power potential but requires design measures to ensure eye safety.
Since blue light-emitting diodes (LEDs), and subsequently white LEDs, were introduced in the mid-1990s, the importance of short-wavelength LEDs for a broad range of applications (such as illumination and medical applications) is becoming increasingly evident.
White LEDs have been used in many applications in the last few years, including indication, LCD, and keypad backlighting; pedestrian signs; automotive dash boards; portable flash light; channel letters; and general illumination. Medical applications also require white light with a high color-rendering index (CRI), enabling users to differentiate between tissues and veins with slight color differences. Specific medical applications include instruments for surgery, endoscopy, and color-picture evaluation. A CRI value of greater than 90 is the target for these applications. Increasingly, users are also showing a preference for so-called unsaturated solid state–based light sources to replace bulb and filter-illumination methods.
Short-wavelength, blue or near-UV, LED chips coupled with phosphor conversion offer solid-state solutions for white lighting as well as for unsaturated colors and some of the saturated colors. Short-wavelength, phosphor-converted LEDs also open the door to new applications, giving users the freedom to choose specific colors or white illumination with high CRI.
White LEDs and unsaturated colors can be generated by exciting phosphors with blue-emitting LEDs to yield emission wavelengths ranging from 510 to 650 nm. Blue indium gallium nitride (InGaN) LED chips, used for this conversion, have a dominant wavelength in the range of 460 nm. Therefore, for colors with a dominant wavelength (λdom) under 510 nm, generated by conversion of blue light, the overlap between excitation and emission spectra significantly reduces the efficiency of the phosphor conversion process. Shifting the excitation wavelength into the near-UV region has helped to remedy this problem. It has also allowed the exploitation of a broad array of phosphors that show an exceptional absorption and quantum efficiency (QE) at UV or near-UV wavelengths, and of the availability of high-power chips for these wavelengths. A drawback of moving into the near-UV, however, is the faster degradation of package materials due to high photon energy that can cause chemical-bond cracks, particularly at higher temperatures, and increase the risk of eye hazards.
Conversion LED efficiencies
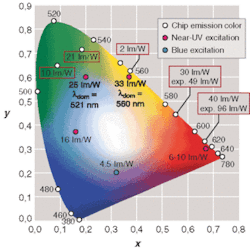
Efficiencies of white phosphor-converted LEDs with short-wavelength excitation have now exceeded 40 lm/W at low currents. But for colors in the range of yellow to red (580 to 650 nm), conversion of short-wavelength LEDs offers no advantage because the LEDs based on semiconductor emission are not only more efficient, but also can be produced at lower costs.
A 49-lm/W wall-plug efficiency can be obtained in the laboratory for yellow (λdom = 590 nm), and values up to 100 lm/W are achieved for red (λdom = 618 nm).
For saturated colors, however, there are still wavelengths for which the efficiencies suffer from material system–based restrictions. In the wavelength range from 530 to 570 nm, for example, the efficiency of the InGaN material system decreases sharply with increasing wavelength. So for pure green (560 nm) to yellowish green (570 nm) wavelengths, the indium gallium aluminum phosphide (InGaAlP) semiconductor system is used. However, efficiencies are as low as 2 lm/W for pure green and approximately 6 lm/W for green (see Fig. 1). Hence, the green wavelength area between about 520 and 570 nm is most suitable for near-UV conversion, because higher efficiencies (compared to the standard chip-based colors) can be expected. For a pure green color, the wall-plug efficiency of the conversion LED is more than 11 times higher than the corresponding InGaAlP-based LED.
Unsaturated colors can be obtained by conversion of blue light using phosphors developed by Osram Opto Semiconductors. For excitation wavelengths in the near-UV range (395 to 410 nm), the absorption and quantum efficiency increase sharply for some of these phosphors. Other phosphors show a high efficiency for near-UV excitation, but do not work with excitation at blue wavelengths (see table, p. 104).
There are also phosphors with emission wavelengths around 490 nm. A conversion LED for this wavelength has a significant advantage, compared to an InGaN-based direct-light emission, because the InGaN-based semiconductor color in this area is strongly dependent on the applied current, yielding a visible color shift from blue to green with changing current.
Near-UV radiation and eye safety
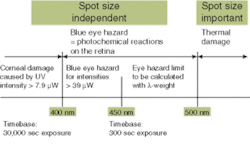
When discussing near-UV excitation, the possibility of eye hazards must be addressed if a significant portion of near-UV radiation is emitted. The standard EN 60825-1 (or IEC 60825-1) describes the classification of lasers as well as LEDs with respect to eye safety and should be taken into account for a standard calculation of the potential hazards. The EN 60825 standard considers lasers and LEDs as equivalent for determining dangerous ratios of radiation emission (see Fig. 2).
The damage mechanisms that are relevant for near-UV LEDs are corneal UV damage and the "blue-light hazard" of potential photochemical damage to the retina over the long term.1 Experience accumulated with conversion using different phosphors has shown that the amount of near-UV remaining power output varies based on the quantum efficiency and absorption level for different phosphors. As quantum efficiency and/or absorption of the excitation wavelength range decrease, the problem of residual emission of near-UV radiation increases.
One technique for minimizing residual power output at the excitation wavelength is the use of a UV-blocking layer. The blocking layer is based on UV-absorbing inorganic particles applied over the LED resin in a multistep casting process. Organic compounds can also be used to attenuate UV radiation. But organic absorbers degrade rapidly with time, and it is not unusual for industrial applications to require lifetimes in excess of 50,000 hours in high ambient temperature and humidity conditions.
Inorganic compounds, such as metal oxides however, can provide efficient UV absorption without chemical degradation over acceptable industrial application lifetimes. The UV-absorbing properties are based upon a steep absorption increase for photons with energies higher than the optical bandgap energy of the inorganic compound. The corresponding wavelength of these photons can be found using the equations: E = hν and ν = c/λ. So photons with an energy bandgap higher than the blocker bandgap have wavelengths shorter than the corresponding wavelength of the blocker and will be strongly absorbed. Titanium dioxide, cerium oxide, and zinc oxide, for instance, are used to provide UV shielding, because they exhibit strong absorption in the near-UV wavelength range and negligible absorption in the visible range.
One important requirement for maintaining high transparency in the visible region, together with efficient blocking of near-UV radiation, is the choice of particle size in the absorbing compound. Usually the Rayleigh criterion for the particle diameter D restricts the particle size. For particle diameters D < l/20, transparency losses due to light scattering can be minimized.
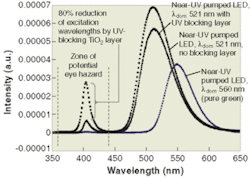
For example, in an LED with the dominant wavelength of 521 nm, the near-UV radiation amounts to about approximately 13% of the total power output (see Fig. 3). A near-UV blocking layer containing nanosized TiO2 on the same LED reduces the amount of excitation radiation emitted to 20 % of the initial intensity. The blocking layer still has potential for improvement, however, because bandgap calculations indicate that the nanosized TiO2 used was a mixture of rutile and anatase TiO2. Substitution of pure rutile TiO2 nanoparticles, for example, would further increase the absorption of near-UV radiation.
REFERENCE
- J. D. Bullough, J. Illuminating Engineering Society 29(2) 141, 6 (Summer 2000).
NADIR FARCHTCHIAN is senior application engineer, LED and Intelligent Display Products, BERT BRAUNE is department headof backend technology for materials and processes, and HERBERT BRUNNER is materials specialist at Osram Opto Semiconductors, 3870 North First St., San Jose, CA 95134; e-mail: [email protected].