Dermatology: Bessel beam works better than Gaussian for skin photothermolysis
Photothermolysis is a way of altering and rejuvenating skin by paradoxically wounding it. Laser light is focused on locations within the epidermis and dermis, causing microscopic burns that eliminate some sort of tissue or other problem and, as they heal, stimulate generation of new skin tissue. The technique can be used to remove port-wine stains and spider veins, as well as tattoos and hair.
The technique requires a tightly focused beam, as the narrower the diameter of the burn volumes, the less extended is the epidermal damage and thus the side effects. Conventional photothermolysis is done using a laser beam with a Gaussian lateral profile and thus a Gaussian focused spot. A group at Philips Research (Eindhoven, Netherlands) is experimenting instead with Bessel beams to increase the depth-to-diameter ratio of the thermal lesions. Bessel beams, which have a Bessel-function cross-section, undergo almost no diffraction over a certain distance, and thus have a columnar focal volume. (However, even a Bessel beam will eventually start to spread—the trick is to design the beam so that the minimally diffractive length matches the application.)
16 mm nonspreading path
Monte Carlo simulations were done along with the experimentation. The simulations produced photon-absorption maps at a 10 μm spatial and 10 μs temporal resolution. A damage-probability threshold of 10% determined the simulation results.
Experimentally, light from a 1.5 W pulsed laser diode operating at a 1435 nm wavelength was passed through an axicon lens with an angle of 5° away from being flat—the resulting Bessel beam was calculated to have a 16 mm nonspreading path in air. For comparison, a Gaussian beam was generated using a lens with an 11 mm focal length, which was calculated to produce a focus with a Rayleigh range (the axial distance at a laser focus within which the beam cross-section no more than doubles in area) of about 50 μm.
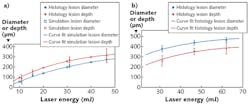
Fresh human skin samples from the abdominal area of donors were used, with the permission of the donors. The samples were each about 5 × 10 mm in area and about 1 mm thick. Eleven lesions were created for each of five different laser pulse energy settings (11, 16, 31, 47, and 62 mJ), for a total of 55 lesions per sample. Experiments were done for both the Gaussian and Bessel beams.
To analyze the results, the researchers cryosectioned the samples, stained them with nitro-blue tetrazolium chloride, and examined them under a microscope. Results showed that the Bessel-beam-produced lesions were generally narrower than those produced using a Gaussian beam (see figure) at equivalent pulse energies-this was the desired result. This indicated that the use of Bessel beams for photothermolysis is best for low pulse energies or short pulse durations.
For Bessel-beam pulse energies <20 mJ, the depth-to-diameter ratio increased as the pulse energy increased. However, above 20 mJ, the depth-to-diameter ratio did not increase much further, approaching a value of 1.17. The researchers speculate that this was a result of the skin's thermal-relaxation response.
In contrast, the depth-to-diameter ratio of the lesions created by the Gaussian beams was less than 1. The difference in depth-to-diameter ratio between the Bessel and Gaussian lesions was one of lesion narrowness rather than depth.
REFERENCE
1. C. Mignon, A. H. Rodriguez, J. A. Palero, B. Varghese, and M. Jurna, Biomed. Opt. Express, 7, 12, 4974–4981 (2016).
About the Author
John Wallace
Senior Technical Editor (1998-2022)
John Wallace was with Laser Focus World for nearly 25 years, retiring in late June 2022. He obtained a bachelor's degree in mechanical engineering and physics at Rutgers University and a master's in optical engineering at the University of Rochester. Before becoming an editor, John worked as an engineer at RCA, Exxon, Eastman Kodak, and GCA Corporation.
