2-D spectroscopy sheds light on photosynthesis
Energy-transfer pathways in the photosynthesis process appear to be affected by molecular factors that had not previously been considered, according to results obtained by researchers at the University of California-Berkeley (UC Berkeley) and Lawrence Berkeley National Laboratory (LBNL; Berkeley, CA). The results are based on direct and highly detailed observations of the process using 2-D spectroscopy.
Researchers often study the photosynthesis process using the Fenna-Matthews-Olson (FMO) photosynthetic protein. The FMO complex is a light-harvesting protein from green sulphur bacteria that serves both as an antenna for collecting light and as a mediator for directing excitation energy from other proteins to the reaction center, where it aids in producing adenosine triphosphate, the “energy currency” in living cells.
“FMO is a model system for studying energy transfer in the photosynthetic process because it is relatively simple (consisting of only seven pigment molecules) and its chemistry has been well characterized,” said Graham Fleming, the lead researcher and the deputy director of LBNL. “As in all photosynthetic systems, the conversion of light into chemical energy is driven by electronic couplings between molecules and we monitored the process as a function of time and frequency.”
Conventional wisdom has held that the molecular energy transfer occurred stepwise down the energy ladder (that is, seven steps for the seven molecular energy levels in FMO). This view was not countered by observations made using conventional time-resolved electronic spectroscopy, which is only capable of directly determining the evolution of populations over time, meaning that the actual coupling between different energy levels could only be inferred indirectly.
Direct and highly detailed mapping of vibrational couplings has become possible, however, with the development of 2-D femtosecond IR spectroscopy based on heterodyne detection of three-pulse photon echoes. The UC Berkeley-LBNL research team has extended this approach into the visible range to directly measure electronic couplings in the FMO molecular complex.
The technique allows scientists to track the transfer of excitonic energy between molecules that are coupled through their electronic states in any photoactive system, macromolecular assembly, or nanostructure. Fleming likened it to the early superheterodyne radios, in which an incoming high-frequency radio signal was converted by an oscillator to a lower frequency for more controllable amplification and better reception. In the 2-D spectroscopy that was applied to the study of electronic couplings in photosynthesis, however, a sample was sequentially illuminated with 50-fs pulses from three laser beams, while a fourth beam was used as a local oscillator to amplify and phase-match the resulting spectroscopic signals.
The 50-fs, 805-nm, 3-kHz laser pulses were produced in the Berkeley experiment using a home-built Ti:sapphire regenerative-amplifier laser system in a diffractive-optic-based noncollinear four-wave-mixing setup with phase-matched box geometry. Movable glass wedges were used to introduce interferometric time delays with a precision on the order of λ/100. Passive interferometric phase stability was maintained over several hours, and the third-order signal was completely characterized by spectral interferometry with the help of a heterodyned local-oscillator pulse, according to the researchers.1 Resolution of individual spectral features was enhanced by performing the experiment at 77 K in a liquid-nitrogen cryostat.
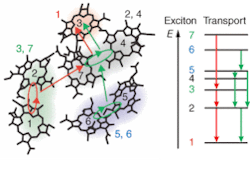
The technique enabled the researchers to follow the energy transfer in time and space as excitons, generated in the FMO pigment by incident photons, propagated between different bacteriochlorophyll molecules. The team noted that most excitonic wavefunctions were delocalized across one or two neighboring molecules. They also noted two major energy transport pathways and that the degree of wavefunction overlap between initial and final energy states on each pathway correlated to skipped rungs on the energy ladder and faster energy transfer (see figure).
“Excitation energy moved through the FMO complex in a smaller number of steps but larger energy increments than was previously supposed,” Fleming said. “What we’re seeing is that nature exploits quantum-mechanical effects by delocalizing excitation energy over two or more molecules in a system.”
Implications for solar power
Because the initial steps in photosynthesis in green plants and cyanobacteria convert sunlight into chemical energy with almost 100% efficiency, the potential implications of unlocking the secrets of photosynthesis for application to solar power generation are significant. “Nature has designed one of the most exquisitely effective systems for harvesting light, with the steps happening too fast for energy to be wasted as heat,” Fleming said. “Current solar-power systems, however, aren’t following nature’s model.”
The surprising experimental results present implications for further study of photosynthesis and broader use of the technique. “This gives us a new way to think about the design of artificial-photosynthesis systems,” Fleming said. “It tells us that we must take into consideration the combined spatial-energetic arrangements of molecules in a system. If the molecules are properly arranged in both space and energy, we can transport energy from one place to another much more efficiently.” The next step in terms of photosynthesis will be to apply the 2-D spectroscopy technique to the study of photosynthesis processes in the reaction center.
Fleming expects that the technique will find broader application, however. “I think this will prove to be a revolutionary method for studying energy flow in complex systems,” he said. “Using 2-D electronic spectroscopy, we can map the flow of excitation energy through space with nanometer spatial resolution and femtosecond temporal resolution.”
REFERENCE
1. T. Brixner et al., Nature434, 625 (March 31, 2005).