One of the biggest challenges in developing optical diagnostic tools has been figuring out how to take complex concepts and state-of-the-art technologies and turn them into easy-to-use clinical devices that can quickly and accurately assess the condition of a wound or the physiology of a patient. In its purest form, the process is simple: shine light onto or into the skin and wait to see what comes back. But making this happen in the blink of an eye is anything but simple.
Still, much progress has been made; consider blood-glucose monitors, pulse oximeters, wavefront analyzers, even optical coherence tomography. Each has become a well-established diagnostic tool in a niche application (or two), paving the way for the next generation of devices aimed at a broad range of point-of-care applications. There are, for example, products on the market (or nearly so) that utilize near-infrared (NIR) spectroscopy for applications ranging from dental imaging to burn diagnostics (see “TruTouch makes alcohol testing a breeze”).
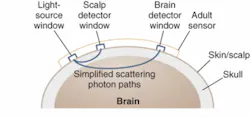
CAS Medical Systems (Branford, CT) recently introduced the FORE-SIGHT cerebral oximeter, which can quantify tissue oxygen saturation by projecting low levels (20 mW) of NIR energy into the brain (see Fig. 1). Protecting the brain from low oxygen levels during surgical procedures is critical, especially for high-risk cardiovascular procedures. While the concept itself is not new, CAS says its latest design takes the accuracy of this technique to a new level.
“From 1977 to the late 1990s, oximeters were trending devices; if you made a calculation that gave a quantitative value, it was based on the trend,” said Paul Benni, senior scientist for CAS Medical. “Later devices came out saying they are more accurate, with the number displayed on the screen indicating the amount of oxygen in the blood. Our system is the first to be approved by the FDA as a device that attaches an absolute value to that number.”
Benni says the company’s proprietary light module and computer algorithms-which have been empirically calibrated on patients-make this possible. FORE-SIGHT operates on the principle that blood contains hemoglobin in two primary forms, oxygenated hemoglobin and deoxygenated hemoglobin, which absorb light in different, measurable ways. Cerebral-tissue oxygen saturation (SctO2) levels are found by determining the ratio of oxygenated hemoglobin to total hemoglobin at the microvascular level.
With sensors placed on the patient’s forehead, laser energy is projected into the brain at four separate wavelengths (690, 780, 805, and 850 nm) to capture information needed for an absolute indication of SctO2 levels. The four wavelengths maximize the measurement accuracy of oxy and deoxy hemoglobin in determining SctO2, compensate for wavelength-dependent scattering losses, and account for interference from other background light absorbers such as fluid, tissue, and skin pigmentation. Reflected light is captured by photodetectors positioned on the sensors; after analyzing this light, the system displays the cerebral-tissue oxygen saturation level on the monitor as an absolute number that is refreshed every two seconds, along with a graph of historical values.
“The fiber optic goes to the sensor, and inside the sensor is a prism that redirects the light for output,” Benni said. “Then you have two p-i-n photodiodes, near and far; the near photodiode tries to cancel out interference from the skin and scalp, while the far photodiode is sensitive primarily to light coming back from the brain to account for all of the light that is scattered.”
Bad plaque versus good
InfraReDx (Cambridge, MA) has developed another FDA-approved clinical device that utilizes NIR spectroscopy for sensing and diagnosis in cardiology-specifically for interventional cardiologists performing cardiac catheterizations (see Fig. 2). The device is designed to distinguish vulnerable, lipid-rich plaques-namely thin-capped fibrous atheromas, which are most likely to rupture and cause a heart attack-from nonvulnerable plaques in coronary arteries. Optical signals obtained from patients during a scan (which typically takes one to two minutes; 5 cm at 1 mm/s) are analyzed with algorithms validated by comparison to tissue histologic findings in ex-vivo specimens and an image called a “chemogram” is produced, indicating the presence of lipid and other chemicals of interest.“Near-infrared spectroscopy is the best way to determine the chemical composition of plaque versus ultrasound that looks at morphological and structure data,” said Bob LaRoche, vice president of sales and marketing at InfraReDx. “Also, our technology can be used in the blood stream without occluding the blood flow.”
The InfraReDx system comprises a tunable diode laser, a 2 m optical fiber (for energy delivery into the arteries), a proprietary 1 mm catheter with an optical tip that delivers and retrieves the NIR light within an artery, a unique pull-back and rotation device that produces a 360° chemical map of the artery, and standard IR photodetectors. The monitor, which sits on a cart, includes the laser source and all the computing hardware.
“Our diode operates in a unique wavelength range, it can tune over the entire bandwidth in less than 10 ms, and it is very linear,” said Jay Caplan, vice president of research and development at InfraReDx. “We are taking spectroscopic measurements at about 100 Hz in a beating heart, so we need a ‘strobe’ approach to freeze the heart motion in one spectrum. By taking about 100 measurements per second we are able to determine the chemical composition of the plaque; each spectrum becomes a single pixel in the chemogram, which helps the physician spatially locate where in the artery the lipid-rich plaque are located.”
Further out
Near-IR spectroscopy is also being studied for burn assessment. Researchers at the National Research Center’s Institute for Biodiagnostics (NRC-IBD; Winnipeg, Man., Canada) have spent the last few years developing and testing a noninvasive NIR imaging tool that will enable doctors to better determine the depth of a burn and the viability of the remaining tissue (see Fig. 3).“At present there is no tool out there to assess a burn other than visual assessment,” said Larry Leonardi, research officer at the NRC-IBD. “Typically a surgeon will look at the burn and based on the appearance decide a course of action. They have criteria, such as whether the tissue is leathery or discolored; they then assess whether it is going to heal or require surgery. If they are unsure, they will wait 24 to 72 hours and then reassess the wound based on what it looks like. We are trying to provide a device to aid in their assessment because they are right about 65% of the time. And if they wait too long (beyond 72 hours) and then try to graft new skin, they face the risk of burn infection or a failed graft.”
The NRC-IBD system utilizes a tungsten halogen light instead of a laser, in part because of the nature of the application; rather than having to go through turbid media such as the skull or down a 2-m-long fiber optic, the photons need only to image about 5 mm into the surface of the skin. In fact, according to Leonardi it is an advantage to have the broader spectral range provided by the lamp; to achieve the same effect with lasers would require a bank of diodes. Other components include a standard back-illuminated CCD camera for detection that is cooled to -20°C and a liquid-crystal filter for wavelength selection.
It takes about 40 seconds to collect a full spectroscopic image series; in its first clinical test, the device was able to distinguish between first- and third-degree burns. Measuring the reflected light can provide information on how much blood is present, the amount of oxygen that is being supplied to the tissue, and the tissue’s moisture content, all sensitive indicators of viability and tissue health.
“We’re looking at tissue oxygenation and tissue blood volume as indicators of whether or not the tissue is going to survive,” Leonardi said.
Another potential application is dental imaging. Here again, while the technology is not yet commercially available, it has been shown to offer several advantages over conventional diagnostic tools used in dentistry, such as x-rays, particularly for early detection of occlusal and interproximal dental decay. According to Daniel Fried, director of the master’s degree program in oral and craniofacial biology at the University of California (San Francisco, CA), dental enamel has been shown to be highly transparent at 1300 nm, and his group has been conducting studies of a 1310 nm NIR transillumination system for imaging early decay.
The system uses 20 mW superluminescent diodes in conjunction with an indium gallium arsenide (InGaAs) focal-plane-array camera fitted with a prism and video lens. The light is beamed through a collimator and cylindrical lens onto an extracted tooth; in studies of occlusal decay, the light was directed just above the gum line of the tooth. The NIR light diffused through the dentin, providing uniform back illumination of the crown enamel and yielding images of the occlusal surfaces (see Fig. 4). According to Fried, the NIR images show high contrast between healthy and demineralized (decay) areas, and demineralization is easily differentiated from stains, pigmentation, and hypomineralization. In addition, the transparency of the enamel enables the detection of decay hidden under the enamel, which often goes unseen on x-rays.TruTouch makes alcohol testing a breeze
Law-enforcement officials are enthusiastic about a new tool for alcohol testing that uses near-infrared (NIR) absorption spectroscopy to determine a person’s blood-alcohol content more swiftly and in a more sanitary way than current methods. The TruTouch 1100 from TruTouch Technologies (Albuquerque, NM), a spinoff of InLight Solutions, is a noninvasive Fourier-transform infrared system that introduces very low-power 1 to 2.5 µm light from a ceramic blackbody source into the skin of the forearm. The reflected light is collected by a single-pixel, thermoelectrically cooled extended InGaAs detector. A HeNe laser from JDSU acts as the internal reference source. The entire process takes about 1 minute, compared to 30 minutes or more for a standard breathalyzer test. The TruTouch can also function as a biometric identity-verification system for, say, prison work-release programs or airline pilots.
“The fact that you don’t have to take any body-fluid sample, including breath, is a big advantage of the TruTouch,” said Ben Ver Steeg, vice president of engineering. “If you had to spend most of the day having intoxicated people spit and breathe at you, this is a surprisingly strong selling point. If a police officer has probable cause for arresting you and gives you a breath test, they have to observe you for 30 minutes to then testify in court that you did not consume alcohol, belch, or vomit because that would change the outcome of the test. The hardest part of this whole thing is that you are measuring people, and people are very different in terms of skin structure, physiology, and so on; they are different enough that we are able to identify them based on their individual optical characteristics.”
About the Author
Kathy Kincade
Contributing Editor
Kathy Kincade is the founding editor of BioOptics World and a veteran reporter on optical technologies for biomedicine. She also served as the editor-in-chief of DrBicuspid.com, a web portal for dental professionals.